Platinum is a very rare metal on Earth, considered precious and malleable for processing. Its name comes from the Spanish language, literally translated as small silver. In the periodic table it appears under the designation Pt. The element is found in nuggets, various alloys and compounds.
Platinum's melting point, heavy weight, hardness and stability in aggressive environments make the metal indispensable in various areas of life.
General characteristics of the metal
Platinum is a noble metal with high density. You can recognize it by its whitish-gray tint and characteristic shine. It does not react to acid, alkali and other elements, which makes it resistant to oxidation and corrosion. The closest neighbors of platinum in the periodic table are iridium, osmium, palladium, rhodium and ruthenium.
The discoverers of platinum were Spanish sailors. Although earlier researchers found artifacts dating back to the times of Ancient Egypt.
When the property of the precious metal to be alloyed with gold was discovered, counterfeit coins and jewelry began to be made in Europe. Yellow metal is also noble and is highly valued, but not as much as platinum.
In nature, platinum is found in admixtures with iron, manganese, nickel and other ores. It can be found in the form of a separate vein or in placers around the mine. There are not so many metal deposits in the world. The main ones are located in South Africa and Russia. There are also deposits in Zimbabwe, Canada, the USA and China.
The precious metal is used in the chemical and industrial sectors, in the automotive industry and astronautics. In medicine, instruments and parts for equipment are made from metal. In addition, platinum molecules have proven themselves in chemotherapy in the treatment of cancer.
Platinum bars
Characteristics and properties of platinum
Platinum has a very high density. Among the heaviest metals, it occupies a special place and is second in weight only to the equally rare ones, iridium and osmium.
Compared to gold and silver, it has greater hardness. It is this property that allows platinum to be resistant to mechanical stress.
The metal is capable of absorbing gases - hydrogen and oxygen. In some conditions this ability is enhanced. Oxygen is absorbed especially well by platinum black, which is obtained as a result of the reaction of saline solutions with reducing agents.
Characteristics:
- metal is white (sometimes with shades of gray), shiny;
- crystal lattice at 20 °C, nm a = 0.392;
- hardness is MPa 390-420;
- electrical resistivity at 0 °C, μΩ•cm 9.85;
- melting point, °C - 1773.5;
- boiling point, °C - 4410;
- atomic radius, nm - 0.138;
- specific heat capacity, J/(mol/K) - 25.9;
- modulus of elasticity, GPa - 173;
- thermal conductivity at 25 °C, W/(m•K) - 74.1;
- density at 20 °C, kg/dm3 - 21.45.
Nuances of processing platinum raw materials
The metal is cleaned of impurities first mechanically, then using chemicals. The latter allows you to improve the quality of the alloy and increase its properties.
Platinum bars are heated in special containers. Aqua regia is also added there. In this way, unnecessary base metals and precipitates containing other elements that cannot be dissolved are separated and dissolved.
The sediment must be filtered and treated with aqua regia. Substances such as osmium and iridium are used for industrial needs, so they are removed from boilers after processing the alloy.
Osmium, palladium and iridium formed during processing must be converted. This is necessary so that ammonium chloride added to the composition cannot affect them, and they do not precipitate. To do this, the solution is changed by heating with sulfuric and oxalic acids.
After ammonium chlorine is introduced, platinum will precipitate in the form of golden granules. It is removed and cleaned with ammonia. The finished product is dried and used for its intended purpose.
Industrial smelting of platinum
Melting methods
When melting precious metals and alloys, it is necessary to create conditions under which their losses are minimized.
Platinum and its alloys are melted in crucible induction furnaces made of calcium oxide, magnesite or zirconium oxide. Melting containers are made of refractory materials. If you need to obtain a product without calcium or magnesium impurities, you can use crucibles made of thorium or zirconium oxides.
Different platinum is used as a raw material for melting: spongy, compressed and in briquettes. You can also use scrap. Additional components are introduced into molten platinum at a temperature of 1850-1900 degrees Celsius. Despite the weak interaction of platinum with furnace gases, smelting must be carried out very quickly, preventing deoxidation of the metal.
Melting platinum at home
Where is it used?
Due to its unique characteristics, platinum is used today in the following industries:
Jewelry industry
Various jewelry is made from this metal; it goes well with many stones (diamonds, gems, etc.). Platinum allows you to reliably protect precious stones, so they do not wear out and retain their elegant appearance for a long time. In addition, this element does not reflect light onto the stone, so it always has a deep and rich color. The most suitable option would be a cut diamond.
Earrings, rings, bracelets, etc. are also made from this noble metal.
All 950 platinum jewelry has a long service life. Wear resistance is not the only advantage of this material, as it has an amazing and very attractive color.
The color of this metal does not darken or fade even after decades, so it is significantly different from gold, which fades and turns yellow over time.
Due to its unique composition, platinum is a hypoallergenic metal, and therefore jewelry made from it can be worn by people with very sensitive skin.
Such jewelry can be found among family heirlooms because they can survive more than one generation.
Medicine
Platinum is in demand in medicine; for example, it is used to produce medical instruments that are not oxidized, but are sterilized during the calcination process, which is often required in extreme field conditions. Electrodes for pacemakers are made from it, and it has also found application in dental prosthetics and hearing aids.
In addition, nanoparticles of the element manage to penetrate the cells of the human body, while they have an anti-inflammatory and regenerating effect, and they also improve the nutrition of cells with useful microelements. Therefore, platinum compounds are now components of many drugs that are intended for cancer patients.
Automotive industry
Due to the excellent catalytic properties of the metal, catalytic converters are now made from it for exhaust gas systems of vehicles, which allow the exhaust gases to be completely burned, leaving behind carbon dioxide and water vapor.
Electrical engineering
The stable electrical and thermoelectric properties of the metal, as well as its anti-corrosion resistance, make it an almost irreplaceable material for electrical engineering and radio electronics.
This metal is necessary for the production of high-precision instruments. It is used to make difficult-to-melt cathodes, anti-cathode X-ray tubes, as well as wire for resistance thermometers, which make it possible to accurately measure temperature over a wide range.
Platinum is used to cover contacts and resistances because it has a high melting point. Powerful magnets are made from an alloy of Pt and Co. This material is necessary for liquid crystal displays, hard drives and other computer parts.
Petrochemical industry
In the petrochemical industry today, platinum mesh is in demand, with which sulfuric acid is produced. Reactor vessels and utensils are made from this metal.
Catalysts from this element make it possible to obtain:
- high octane gasoline;
- aromatic hydrocarbons;
- technical hydrogen.
Glass industry
Filters are made from this metal, which are necessary for drawing glass alloys. In such crucibles, optical glass is melted if it is necessary to maintain the required recipe. In addition, this metal is used in laser mirrors.
Banking sector
Coins are minted from platinum and its alloys. The first coins were minted in Russia in 1828. Today such coins are considered to be investment coins.
In addition, this noble metal is used to cast ingots, which are used for investment.
The demand for platinum among bankers, scientists, jewelers and technicians is increasing every day, and therefore the value of the metal is increasing, which makes it very attractive to investors. Further study of the metal will further expand the scope of its application.
Melting temperature
Platinum, as an individual element, has a melting point of 1774 degrees. But this indicator may vary depending on the content of alloys in the alloy. For example:
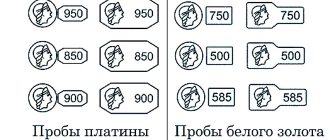
- platinum alloy with iridium 850/900/950 standard - 1800-1820/1780-1800/1780-1790 degrees;
- platinum alloy with palladium 850/900/950 standard - 1730-1750/1740-1755/1755-1765 degrees;
- alloy with 950 cobalt - 1765-1780 degrees;
- platinum alloy with 950 ruthenium - 1780-1795 degrees.
The melting point of platinum and its other physical characteristics make it possible to use the metal in the production of viscose fiber. Special high-strength molds are made from the alloy, through which plastic material is pressed.
The melting point of the precious metal must be taken into account when introducing alloy components into its composition during the formation of jewelry materials.
The story about counterfeiters and Ural hunters
The cheap “silver” gave the counterfeiters a great idea. The annoying metal was added to gold: the color of the alloy did not change, and the fakes were of high quality.
The Spanish crown, having learned about the tricks of the scammers, ordered the “harmful metal” to be drowned. Naturally drown in the waters of the Atlantic - in Colombia and Spain. Later, the Spanish court decided to make some money on the “rotten silver” and add it to coins.
The sad story of the “metal Cinderella” ended by the 19th century.
Scientists became interested in platinum and discovered that it has a “family.” Together with the “head” there are five family members. These are rhodium, iridium, ruthenium, palladium, osmium. The classification was carried out by scientists from different countries.
Interesting: Ural hunters, encountering platinum placers, used metal grains instead of shot. It was the most expensive shot in the world.
Melting platinum at home
If a situation arises when you need to melt platinum at home, you first need to familiarize yourself with this process. Before the procedure, prepare the necessary tools and materials. Platinum can be melted using homemade gas burners using oxygen cylinders. A welding machine is also used for these purposes. A porcelain crucible is used as a container.
Laboratory porcelain crucible
The mixture is prepared in the same way as in industrial conditions. The melting process itself is performed as follows.
- The platinum alloy is placed in a crucible and heated with a gas burner or other homemade devices until the required temperature is reached.
- The type of metal cast depends on the mold created and personal preference.
- After melting, the platinum must cool completely.
- The finished product is removed from the mold using tools and taking the necessary precautions.
As you can see, you can melt platinum yourself, although the final result largely depends on the skills of the master, the quality of materials and knowledge of technology. It is almost impossible to obtain a pure product without impurities without special equipment, because... All melting methods are not ideal. If you need pure metal, it is better to contact specialists.
History of discovery
The Sandia National Laboratories have created the most durable and wear-resistant material. It was designed at the atomic level using a computer. Gold and platinum combined in the alloy strengthened each other's technical characteristics and added very high wear resistance, 100 times higher than high-alloy steels.
Electron microscope studies have shown that when the components are mixed, the unique metal alloy forms a thin layer of diamond lubricant on its surface. Thanks to it, the abrasion resistance increases several times and is superior to high-alloy metals and bronzes.
The alloy was called white gold because it has a dazzling steel luster with a light, barely noticeable tint of yellow. The main component of the alloy is platinum, the percentage of which reaches 90%. And only a tenth is gold.
Why is gold melted and mixed with less precious metals?
Gold is a hard, refractory metal that is often mixed with other metals such as copper, silver, iron, aluminum, and platinum. At the same time, it often changes color (it becomes white, brown, red). Purposes of alloys:
- Increased strength. Pure gold is easily deformed. Therefore, it is fused with stronger elements - this way you can get a harder material.
- Saving money. Pure gold is an extremely expensive metal. To save money, it can be fused with cheaper components - the price of the final product will be lower.
- Giving gold a unique color (pink, white, green, blue, purple and other alloys). Ligatures of various metals are responsible for the corresponding shade.
To mix metals, they need to be heated to a certain temperature at which it turns from solid to liquid (melting phenomenon). This indicator is called the melting point. If necessary, the material can be heated even after it has melted.
Once a certain temperature is reached, gold may begin to evaporate (this point is called the boiling point).
Gold bullion production process
Equipment and materials for home metal smelting
The list of necessary equipment depends on the method of metal remelting:
- Melt using a burner. To carry out the work you will need a gas cylinder and a burner (autogen). It is recommended to melt gold on a refractory brick, which will not begin to crumble when heated. The crucible can be made from refractory clay. It is also recommended to use refractory tongs to place the material into the crucible.
- Melt using a melting furnace. Most melting furnaces sold in online stores are in the jewelry category. They are completely autonomous (the design contains its own crucible). Therefore, you will only need gold particles.
- Melt using a microwave. For melting, use a microwave oven with a power of 1200 watts or more. You will also need two bricks - two recesses should be made in them (the bricks are placed in the oven with the recesses facing each other to create free space). It is recommended to make the crucible from graphite (microwave radiation can melt the clay).
How to care for white gold jewelry
The precious metal alloy does not oxidize. But white gold products attract dust. Connecting with air moisture, it is firmly held on the surface and clogged into the holes. Jewelry should be cleaned once a month.
- Make a solution of regular soap with a minimum amount of additives. “Children’s” and “Household” are best suited.
- Place jewelry in it for a few minutes.
- Remove and wipe with a soft cloth or paper towel.
- The easiest way to clean a textured surface is with a soft-bristled toothbrush.
Jewelry is coated with a thin layer of rhodium during production. It enhances the shine of precious metals. Hard rhodium provides durable protection against scratches and abrasions. Gradually, the protective coating is worn out in places and should be renewed. You won't be able to do this on your own. You should take your jewelry to a jeweler for cleaning.
The microcircuits are wiped with a damp cloth when the equipment is turned off. After this they are dried.
White gold is the name given to various alloys in which alloys are added to the precious metal to whiten it and improve its technical characteristics. The wear resistance of the metal justifies its high cost.
Composition and structure
When creating an alloy, gold and platinum go through 2 stages of solution formation. First, the molten metals dissolve into each other at the molecular level. Upon cooling, a solid solution is formed when crystals of a pure substance are mixed in similar solidified formations of another component.
Beginners often refer to white gold as:
- silver;
- platinum;
- a separate grade of white gold.
In fact, jewelers at the beginning of the 20th century began mixing 9 parts gold with 1 part palladium. This amount of ligature is enough for the material to acquire a white color and a bright steel shine. This was done in order to pass it off as platinum, which, due to its high melting point, was produced in small quantities and was very expensive. Currently, both precious metals have the same value.
Gradually, other “white gold” compositions began to be produced. The cheapest was an alloy of gold and nickel. The composition had high hardness and was well polished. The lack of material was discovered later. The nickel content in the alloy, even within 10%, causes allergies in some people upon contact with skin, especially sweaty or covered with creams.
Silver as a ligature turns gold white, but the surface is matte.
In the jewelry industry, ligatures made from other materials are used.:
- cadmium;
- platinum;
- zinc;
- copper
They are used to make a two-component and complex alloy. It is enough to add 8.4% platinum to make gold white. Copper, on the contrary, makes it red. Cadmium reduces the melting point of a mixture of noble metals, increases ductility and malleability.
Zinc increases the fluidity of gold, allowing it to fill complex shapes well. In small quantities, zinc whitens the precious metal. If it exceeds 5%, a greenish tint and fragility appear.
Nickel ingots
Advantages and disadvantages
Just look at diamond jewelry to be convinced that white gold serves as a magnificent setting for them. Jewelry can be worn day or evening.
Yellow gold loses its shine faster. It is softer, constantly scratches and oxidizes over time. In jewelry with diamonds, the yellow metal imparts its hue to them, changing the play of light in the stone. It is superior to alloy when manufactured by casting because it has a relatively low melting point and high ductility.
The cost of platinum and gold is approximately the same. Therefore, precious metals in pure form and alloy are equally expensive. When struck by a hard object they ring melodiously. The sound fades away in waves.
Hallmark on 585 hallmark
The authenticity of the yellow metal must always be confirmed by a stamp indicating its purity. In Russia, this procedure is mandatory and controlled by the state. The hallmark allows the buyer to be confident in the quality and authenticity of the precious metal.
The modern laser method allows you to place a mark with a high degree of readability. But the image can quickly be erased, especially if the branding site is constantly in contact with another surface. The mechanical method is still recognized as the most durable.
An example of a mark on a product and its explanation.
The mark itself consists of three parts: a letter (the hallmark code), a drawing (a woman’s head with a kokoshnik, looking to the left), and the number “585” (the hallmark itself). In addition to these symbols, a piece of jewelry may have another mark – the manufacturer’s name.
Products that are imported into Russia are also tested. In Italy and the USA there is no state standard and all responsibility for the authenticity of jewelry lies only with the manufacturer. The mark on foreign products usually consists of the symbols “14 K” (14 carats, corresponding to the domestic 585 standard) and letters indicating the manufacturer.