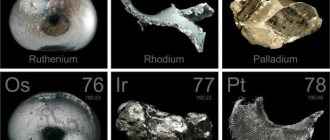
Being in nature
The iridium content in the earth's crust is negligible (10−7% by mass). It is much rarer than gold and platinum. It is found together with osmium, rhodium, rhenium and ruthenium. Refers to the least common elements. Iridium is relatively common in meteorites. It is possible that the actual content of the metal on the planet is much higher: its high density and high affinity for iron (siderophilicity) could lead to the displacement of iridium deep into the Earth, into the core of the planet, during its formation from the protoplanetary disk. A small amount of iridium has been discovered in the photosphere of the Sun.
Iridium is found in minerals such as nevyanskite, sysertskite and aurosmiride.
Iridium mining
Despite the high value of the metal, its quantity on our planet is negligible. Even valuable platinum cannot compete with its rarity. In nature, in the form of nuggets, it is most often found with impurities of other metals, including ruthenium, osmium, palladium or platinum. In combination with these metals, iridium forms aurosmirides, nevyanskites, osmiridium, and sysertskites. Iridium placers can be found in copper and nickel mining areas. It is also possible to extract it from gold deposits.
The leading producing countries are Canada, the USA, South Africa, the island of Tasmania, and New Guinea. The main supplier of iridium to the world market is South Africa.
How much iridium South Africa produces, as well as the rise or fall in demand for the metal, plays a big role in metal mining.
Ruthenium $17 per gram
The metal is named after Russia (from the Late Latin word Ruthenia - Russia). This is also a hard and at the same time very brittle metal, the rarest of the platinum group. It is used in the manufacture of wires, contacts, electrodes, laboratory glassware, and jewelry. In Western Europe and Japan, more and more metal is used for the production of resistors and printed circuits in the electronics industry, as well as for the production of chlorine and various alkalis. This metal comes entirely from South Africa.
Mining locations
The chemical element is found in alloy form in folded earth rocks of the mountains of Russia, peretonite rocks located in South Africa, Kenya, South America, etc.
Where there is platinum, there is also iridium.
About the characteristics of metal as a chemical element:
Characteristic Designation, meaning
| Iridium is represented by the symbol | Ir |
| Number in the periodic table | 77 |
| Atomic weight | 192.22 amu |
| Oxidation states | From 1 to 6 (5 not included) |
| Density at room temperature | 22.7 g/cm^3 |
| Density in liquid state | 19.39 g/cm^3 |
| Melting | At 2300 degrees Celsius |
| Boiling of liquid iridium | At 45 degrees Celsius |
| Has a crystal lattice | Face-centered cube |
The element is found in different colors, the most common is white - KIrF6, lemon - IrF5, gold - K3IrCl6, light green - Na3IrBr6, pink - Cs3IrI6, crimson - Na2IrBr6, dark blue - IrI3. The variety of colors is due to the presence of various salts in iridium.
By the way, the metal got its name due to this variety of colors. Iris is the goddess of the rainbow in Greek mythology.
Receipt
The main source of iridium is anode sludge from copper-nickel production. Au, Pd, Pt, etc. are separated from the concentrate of platinum group metals. The residue containing Ru, Os and Ir is fused with KNO3 and KOH, the melt is leached with water, the solution is oxidized with Cl2, OsO4 and RuO4 are distilled off, and the precipitate containing iridium is fused with Na2O2 and NaOH, the melt is treated with aqua regia and a solution of NH4Cl, precipitating iridium in the form of (NH4)2[IrCl6], which is then calcined to obtain metallic Ir. A promising method for extracting iridium from solutions is the extraction of hexachloroiridates with higher aliphatic amines. The use of ion exchange is promising for separating iridium from base metals. To extract iridium from minerals of the iridium osmide group, the minerals are fused with BaO2, treated with hydrochloric acid and aqua regia, OsO4 is distilled off, and iridium is precipitated in the form of (NH4)2[IrCl6].
How do you get it?
We have already said that this metal is found only in alloys. But how is it possible to obtain iridium? The source of the rock is anode sludge from copper-nickel production. The product - sludge is saturated, after which, under the influence of “regia vodka”, it is transferred from a solid to a liquid state, in the form of chloride compounds H2[PtCl6].
As a result, chemists obtain a liquid mixture of metals and add ammonium chloride NH4Cl to it. After this, the precipitate is removed from the platinum, and then an iridium complex (NH4)2[IrCl6] is obtained. (NH4)2[IrCl6] is calcined with oxygen and nitrogen. The output is metallic iridium.
Properties of iridium
The physical properties of iridium are quite impressive. It is a very hard, heavy metal that is difficult to machine. The melting point is 24660C, and has a fairly high boiling point of 44280C. Its hardness is determined by its density - 22.65 g/cm3. It is stable when heated and at ordinary temperatures; at temperatures up to 1000C it does not react to all known acids. In the presence of alkali metal chlorides at temperatures of 600-9000C, iridium powder can be dissolved by chlorination. It interacts with F2 when the temperature reaches 4500C. The properties of iridium do not play any biological role; it is not a toxic metal, although some of its compounds are very poisonous.
Iridium indicators:
- alloys of iridium with tungsten and thorium are used as metal for thermoelectric generators; with other metals they make fuel tanks for spacecraft, thermocouples, and thermionic cathodes;
- in the automotive industry they are used in spark plugs, which allows them to be used for a long time, although they are expensive;
- finds its application in the manufacture of nibs for ink pens, on gold nibs;
- iridium-192 is successfully used in flaw detection, where generating sources cannot be used, for example, in an explosive environment;
- It is interesting that the kilogram standard is made from iridium, since the alloy of iridium with platinum has mechanical strength and does not oxidize;
- is used in the jewelry industry, but the price for such jewelry is incredibly high;
- make laboratory crucibles to conduct experiments with fluorine, as well as its aggressive compounds;
- make highly durable, heat-resistant glass blowing mouthpieces.
Materials and methods for development and optimization
When conducting experiments, scientists usually take pure iridium obtained from a cylindrical rod through electron beam zone melting. The iridium purity level is 99.95%. The density of the material is noted at 22.37 g/cm3, while the relative density reaches 99%, respectively.
The micro-hardness of the structures under study must have a value of at least 450. The table below shows the concentrations of impurities characteristic of the iridium test samples. Cylindrical samples are cut from forged iridium rod and machined to final dimensions of 6 and 9 mm in diameter and length, respectively.
| Chemical element | Parts per million (ppm) |
| Platinum (Pt) | 30 |
| Ruthenium (Ru) | 30 |
| Rhodium (Rh) | 10 |
| Palladium (Pd) | 10 |
| Gold (Au) | 10 |
| Silver (Ag) | 10 |
| Copper (Cu) | 10 |
| Iron (Fe) | 10 |
| Nickel (Ni) | 10 |
| Silicon (Si) | 10 |
Hot compression testing procedure
The methodology for experiments using the Gleeble-1500D tester seems to be quite successful. Testing of the deformation behavior of iridium is carried out here under thermal compression in the temperature range of 1200-1500°C and taking into account the range of strain rates from 10-1 s-1 – 10-3 s-1.
Under vacuum conditions, the samples are preheated to the compression temperature taking into account a heating rate of 20°C/s and maintained at this temperature for 60 s. Then they are brought to true deformation - 0.6.
Scheme of hot compression tests of pure iridium: 1 – deformation temperatures; 2 – heating rate; 3 – holding time; 4 – strain rate; 5 – hardening with water; T—temperature axis; B - time axis
The temperature deviation of the tester is acceptable within 10°C. Deformed samples are quenched to preserve the high-temperature deformed microstructure for later analysis. To study the hardness profile of compressed samples, a tungsten indenter equipped with a spherical tip is used.
To reduce friction and increase uniformity of deformation during thermal compression tests, sheets of graphite and tantalum are used. The temperature-time diagram of the thermal compression test is shown in the picture above.
Characteristics of the microstructure of iridium metal
Samples for microstructural analysis are cut from the central region of the deformed samples in the direction of compression. Next, the samples are ground and etched using electrolytic technology using 100 ml of a saturated NaCl solution containing 35 grams of H and 3–5 grams of HCl.
Electrical corrosion apparatus: 1 – test iridium; 2 – wall of a laboratory flask; 3 – electrolyte; 4 – electrode based on a graphite rod; 5 - ammeter
The apparatus used for electrolytic corrosion is shown in the picture above. Precision instruments SEM TM4000PLUS, optical microscopy PMG3 manufactured by Olympus are also used. Using this kit, the deformation microstructure is observed and analyzed.
Using information: MDPI
Physical properties
Iridium is a heavy, silvery-white metal that, due to its hardness, is difficult to machine. Melting point - 2739 K (2466 °C), boils at 4701 K (4428 °C). The crystal structure is face-centered cubic with a period a0=0.38387 nm; electrical resistance - 5.3⋅10−8 Ohm m (at 0 °C), and 2⋅10−7 Ohm m (at 2300 °C); coefficient of linear expansion - 6.5⋅10−6 degrees; normal elastic modulus - 538 GPa; density at 20 °C - 22.65 g/cm³, liquid iridium - 19.39 g/cm³ (2466 °C).
Isotopic composition
Natural iridium occurs as a mixture of two stable isotopes: 191Ir (content 37.3%) and 193Ir (62.7%). Radioactive isotopes of iridium with mass numbers 164-199, as well as many nuclear isomers, have been obtained by artificial methods. Artificial 192Ir has become widespread.
Chemical properties
Iridium is stable in air at ordinary temperatures and heating; when the powder is calcined in a stream of oxygen at 600–1000 °C, it forms a small amount of IrO2. Above 1200 °C it partially evaporates in the form of IrO3. Compact iridium does not react with all known acids and their mixtures at temperatures up to 100 °C. Freshly precipitated iridium black partially dissolves in aqua regia to form a mixture of Ir(III) and Ir(IV) compounds. Iridium powder can be dissolved by chlorination in the presence of alkali metal chlorides at 600–900 °C or by sintering with Na2O2 or BaO2 followed by dissolution in acids. Iridium interacts with F2 at 400–450 °C, and with Cl2 and S at red heat.
Oxidation state of iridium
Iridium atoms in compounds have oxidation states of 6, 5, 4, 3, 2, 1, 0, -1.
The oxidation state is the conditional charge of an atom in a compound: the bond in a molecule between atoms is based on the sharing of electrons, thus, if an atom’s charge virtually increases, then the oxidation state is negative (electrons carry a negative charge), if the charge decreases, then the oxidation state is positive.
Iridium ions
6+Ir 5+Ir 4+Ir 3+Ir 2+Ir 1+Ir
Ir 01-Ir
Valency Ir
Iridium atoms in compounds exhibit valence VI, V, IV, III, II, I.
The valency of iridium characterizes the ability of the Ir atom to form chemical bonds. Valence follows from the structure of the electron shell of the atom; electrons involved in the formation of chemical compounds are called valence electrons. A more extensive definition of valence is:
The number of chemical bonds by which a given atom is connected to other atoms
Valence has no sign.
Quantum numbers Ir
Quantum numbers are determined by the last electron in the configuration; for the Ir atom these numbers have the value N = 5, L = 2, Ml = -1, Ms = -½
Trivalent iridium compounds
- Ir2O3 is a dark blue solid. Slightly soluble in water and ethanol. Dissolves in sulfuric acid. It is obtained by lightly calcining iridium(III) sulfide.
- IrCl3 is an olive green volatile compound. Density - 5.30 g/cm³. Slightly soluble in water, alkalis and acids. At 765 °C it decomposes into IrCl2 and chlorine, at 773 °C into IrCl and chlorine, and above 798 °C into its constituent elements. It is obtained by the action of chlorine on iridium heated to 600 °C.
- IrBr3 - olive green crystals. Soluble in water, slightly soluble in alcohol. Dehydrates when heated to 105-120 °C. When heated strongly, it decomposes into elements. Prepared by reacting IrO2 with hydrobromic acid.
- Ir2S3 is a brown solid. Decomposes into elements when heated above 1050 °C. Slightly soluble in water. Dissolves in nitric acid and potassium sulfide solution. It is obtained by the action of hydrogen sulfide on iridium (III) chloride or by heating powdered metal iridium with sulfur at a temperature not exceeding 1050 °C in a vacuum.
Quadrivalent iridium compounds
- IrO2 are black tetragonal crystals with a rutile-type lattice. Density - 3.15 g/cm³. Slightly soluble in water, ethanol and acids. Reduced to metal by hydrogen. Thermally dissociates into elements when heated. It is obtained by heating powdered iridium in air or oxygen at 700 °C, heating IrO2*nH2O.
- IrF4 is a yellow oily liquid that decomposes in air and is hydrolyzed by water. melting point 106 °C. Prepared by heating IrF6 with iridium powder at 150 °C.
- IrCl4 is a hygroscopic brown solid. It dissolves in cold water and decomposes with warm (water). It is obtained by heating (600–700 °C) metallic iridium with chlorine at elevated pressure.
- IrBr4 is a blue substance that diffuses in air. Soluble in ethanol; in water (with decomposition), dissociates when heated into elements. It is obtained by reacting IrO2 with hydrobromic acid at low temperature.
- IrS2 is a brown solid. Slightly soluble in water. It is obtained by passing hydrogen sulfide through solutions of iridium (IV) salts or by heating powdered metal iridium with sulfur without access of air in a vacuum.
Hexavalent iridium compounds
- IrF6 - yellow tetragonal crystals. melting point 44 °C, boiling point 53 °C, density - 6.0 g/cm³. Under the influence of metallic iridium it turns into IrF4, and is reduced by hydrogen to metallic iridium. It is produced by heating iridium in a fluorine atmosphere in a fluorite tube. A very strong oxidizing agent that can oxidize even water:
2IrF6 + 10H2O = 2Ir(OH)4 + 12HF + O2, or NO: NO + IrF6 = NO+[IrF6]-
- IrS3 is a gray, slightly water-soluble powder. It is obtained by heating powdered metal iridium with excess sulfur in a vacuum. Strictly speaking. is not a compound of hexavalent iridium, since it contains an SS bond.
- Ir(OH)4 (IrO2*2H2O) is formed by neutralizing solutions of chloroiridates(IV) in the presence of oxidizing agents. The precipitate Ir2O3*nH2O precipitates when chloroiridates (III) are neutralized with alkali and is easily oxidized in air to IrO2. Practically insoluble in water.
Rainbow metal
The metal was discovered and given its name by the Englishman S. Tennant, a chemist. While conducting experiments with native platinum, the chemist also examined the solutions remaining from the experiments. And not in vain - in them he discovered multi-colored salts of an unknown element.
Iridium was named after the rainbow (in Greek iris) - a multi-colored miracle.
This is not about the color of the metal, it is about the variety of colors of iridium salts:
- K3IrCl6, IrF6 - golden yellow crystals;
- KIrF6 - white crystals;
- Ir2O3 - blue, blue-black crystals:
- IrCl2 - green crystals;
- Na2IrBr6 - crimson color.
There are iridium compounds colored olive, brown, pink, and golden.
Peculiarities
It’s worth saying right away that iridium is a metal. Therefore, it has all those properties that are typical of other metals. Such a chemical element is designated by a combination of the Latin symbols Ir. In the periodic table it occupies the 77th cell. The discovery of iridium occurred in 1803, as part of the same research in which the English scientist Tennant isolated osmium.
The starting material for the production of such elements was platinum ore delivered from South America. Initially, the metals were isolated in the form of a sediment, which “regia vodka” did not “take.” The study revealed the presence of several previously unknown substances. The element received its verbal designation because its salts look as if they shimmer with a rainbow.
The content of iridium in nature is extremely low, and it is one of the rarest substances on Earth.
Chemically pure iridium does not have any rainbow color. But it is characterized by a rather attractive silver-white color. Toxic properties have not been confirmed. However, certain iridium compounds can pose a danger to humans. Fluoride of this element is especially poisonous.
A number of Russian and foreign enterprises are engaged in the production and refining of iridium. Almost the entire output of this metal is a by-product of processing platinum raw materials. Although iridium is not purple, it contains 2 isotopes naturally. Elements 191 and 193 are stable. But a number of artificially produced isotopes have pronounced radioactive properties; their half-life is short.
[edit] Links
- Iridium at Weelements.com.
Electrochemical activity series of metals
, , , , , , , , , , , , , , , , , , , , , , , , , , , , , , , , , , , , , , , , , , , , , , , , , , , , , , , H2
, , , , , , , , , , , , , , ,
Ir, ,
| Periodic table of chemical elements by D. I. Mendeleev | |||||||||||||||||||||||||||||||||||||||||||
| Ir | |||||||||||||||||||||||||||||||||||||||||||
| Uue | Ubn | Ubu | Ubb | Ubt | Ubq | UBP | Ubh | ||||||||||||||||||||||||||||||||||||
| Alkali metals | Alkaline earth metals | Lanthanides | Actinoids | Superactinoids | Transition metals | Other metals | Semimetals | Other non-metals | Halogens | Noble gases | Properties unknown | ||||||||||||||||||||||||||||||||
Application
Of particular interest as a source of electricity is its nuclear isomer, iridium-192m2 (half-life 241 years).
Alloys with W and Th are materials for thermoelectric generators, with Hf are materials for fuel tanks in spacecraft, with Rh, Re, W are materials for thermocouples operated above 2000 °C, with La and Ce are materials for thermionic cathodes.
Iridium is also used to make pen nibs. A small ball of iridium can be found on the tips of nibs and ink rods, and is especially visible on gold nibs, where it is a different color from the nib itself.
Iridium in paleontology and geology is an indicator of the layer that formed immediately after the fall of meteorites.
Iridium, along with copper and platinum, is used in spark plugs of internal combustion engines (ICE) as a material for the manufacture of electrodes, making such plugs the most durable (100-160 thousand km of vehicle mileage) and reducing the requirements for sparking voltage. The first company to use iridium, thereby improving the quality of spark plugs, was the Japanese company NGK. Initially used in aviation and racing cars, then, as the cost of production decreased, it began to be used in mass-produced cars. Currently, such spark plugs are available for most engines, but are the most expensive.
Iridium-192 is a radionuclide with a half-life of 74 days, widely used in flaw detection, especially in conditions where generating sources cannot be used (explosive environments, lack of supply voltage of the required power).
Platinum-iridium alloy has great mechanical strength and does not oxidize. In particular, the kilogram standard is made from this alloy.
In 2013, iridium was used for the first time in the world in the production of official coins by the National Bank of Rwanda, which issued a coin made of pure metal of 999 purity. An iridium coin was issued in the denomination of 10 Rwandan francs.
Industry
The properties of the metal determined the areas of application in the industrial complex:
- Iridium is used to make dishes, equipment, and catalysts for chemical enterprises.
- Single crystals are grown in iridium crucibles.
- In the automotive industry, this is the material of the electrodes (with copper and platinum) of spark plugs. They last longer and are used for luxury cars.
- Iridium-ruthenium alloys contain electric generators and thermocouples for measuring temperatures up to 2000°C.
- This is an indicator of the quality of welds made of steel and aluminum alloys.
The kilogram and meter standards are made from a platinum-iridium alloy (located in Paris).
The third millennium opened up space prospects:
- American Elements has created a technology for casting seamless iridium rings for use on satellites and spacecraft (2006).
- Metal is present in the displays of computers, televisions, iPhones, and other gadgets based on organic light-emitting diodes (OLED technology).
The nuclear isomer of iridium-192 with a “lifetime” of more than 240 years is considered as an energy source.
Medicine
Surgical instruments and parts of pacemakers are made from an alloy of platinum and iridium.
This is the basis for the development of nuclear medicine. Radionuclide raw materials are produced in nuclear reactors and cyclotrons. 90% goes for export.
Russia is one of the world's top five producers of raw medical isotopes.
Iridium-192 is used in flaw detection and oncologists (gamma knife).
Fabulous prospects for iridium in medicine
Pacemakers use an iridium-platinum alloy.
Oncologists use the iridium isotope Ir-192 as a source of gamma radiation. It is used to treat breast and prostate cancer (in the early stages of the disease).
Methods have been developed for the treatment of epilepsy, Parkinson's disease, and schizophrenia by introducing iridium electrodes into the brain. The method of implanting microelectrodes opens up bright prospects for creating prosthetic eyes and hearing aids.
Jewelry
Jewelers value the metal for its strength, using it in an alloy with platinum.
The American company Smithson Tennant was the first to launch the production of iridium jewelry. Iridium wedding rings are positioned by her as eternal in the literal sense: even concentrated acid and time are powerless. They do not require special care or storage conditions.
Jewelry made from alloys with the addition of iridium still looks like it was just purchased after decades.
Jewelry with iridium (even wedding rings) does not wear out and always looks like new. This advantage is especially important for products that experience increased loads (wedding rings, rings, bracelets).
Other areas
Iridium is revered by geologists and paleontologists: it is a marker of the age of layers of the earth's crust and extinct organisms.
The only coin in the world - iridium fineness 999 - was issued in 2013 by the National Bank of Rwanda (a state in eastern Africa). The denomination was 10 local francs.
Until the 1980s, iridium alloy balls were found in the nibs of luxury fountain pens, including the legendary Parker 51. It is an attribute of the elite and an accessory for the luxury fan of James Bond.
California-252 – $6,500,000 per gram
The world reserve of californium is a few grams, probably no more than 5 g. On Earth, only 2 reactors can produce it. One reactor is in Russia, the other is in the USA. Each of the reactors produces 20–40 micrograms per year. California is incredibly expensive. What properties, despite this, make this isotope so necessary?
California-252 has a half-life of 2.6 years. In this case, 3% of all atoms spontaneously fission and with each fission four neutrons are released. 1 g per second releases 2.4 billion neutrons. This corresponds to the neutron flux of an average nuclear reactor! If one wanted to obtain such neutron radiation in the classical way from a radium-beryllium source, then this would require 200 kg of radium. Such a huge reserve of radium does not exist on Earth at all. Even such an invisible amount as 1 microgram of California-252 produces more than 2 million neutrons per second. Therefore, californium-252 has recently been used in medicine as a point source of neutrons with a high flux density for local treatment of malignant tumors.
Biological role
Does not play any biological role. Iridium metal is non-poisonous, but some iridium compounds, such as its hexafluoride (IrF6), are very poisonous.
Iridium - element 77 of the periodic table
The atomic mass of element No. 77 is 192.2. In the periodic table it is located between osmium and platinum. And in nature it is found mainly in the form of iridium osmide, a frequent companion of native platinum. There is no native iridium in nature.
Iridium is a silvery-white metal that is very hard, heavy and durable. According to the data, this is the heaviest element: its density is 22.65 g/cm3, and the density of its constant companion, osmium, the second heaviest, is 22.61 g/cm3. True, most researchers adhere to a different point of view: they believe that iridium is still a little lighter than osmium.
The natural property of iridium (aka platinoid!) is high corrosion resistance. It is not affected by acids either at normal or at elevated temperatures. Even the famous aqua regia is too tough for monolithic iridium. Only molten alkalis and sodium peroxide cause oxidation of element No. 77.
Iridium is resistant to halogens. It reacts with them with great difficulty and only at elevated temperatures. Chlorine forms four chlorides with iridium: IrCl, IrCl2, IrCl3 and IrCl4. Iridium trichloride is most easily obtained from iridium powder placed in a stream of chlorine at 600°C. The only halide compound in which iridium is hexavalent is fluoride IrF6. Finely ground iridium is oxidized at 1000°C and in a stream of oxygen, and depending on the conditions, several compounds of different compositions can be obtained.
Like all platinum group metals, iridium forms complex salts. Among them there are also salts with complex cations, for example [Ir(NH3)6]Cl3 and salts with complex anions, for example K3[IrCl3] · 3H2O. As a complexing agent, iridium is similar to its neighbors on the periodic table.
Pure iridium is obtained from native osmic iridium and from the remains of platinum ores (after platinum, osmium, palladium and ruthenium have been extracted from them). We will not dwell on the technology for producing iridium, referring the reader to the articles “Rhodium”, “Osmium” and “Platinum”.
Iridium is obtained in the form of a powder, which is then pressed into semi-finished products and fused, or the powder is melted in electric furnaces in an argon atmosphere. Pure iridium can be forged when hot, but at normal temperatures it is brittle and cannot be processed in any way.
Kill Cancer: Iridium, Human Serum Albumin and a Little Blue Light
One of the most famous Marvel superheroes has always been and will be considered Logan, aka Wolverine. And what feature of his body comes to mind first, besides, of course, regeneration at the speed of the Flash? In a word - adamantium. This rare metal has unique properties; it is almost impossible to destroy it, and recycling takes a lot of effort. This fictional substance has several equivalents in our reality, which also have very specific properties. Among them, iridium has received special attention from scientists. This metal can hardly make a superhero out of an ordinary person, but it can destroy cancer cells (Deadpool would not refuse this). How did scientists come to this conclusion, how effective is iridium in the fight against cancer and what is its future in oncology? Let's dive into the research group's report for answers. Go.
Metallurgy
Iridium (Ir) is an extremely hard transition metal from the platinum group. Like the fictional adamantium, iridium is very resistant to corrosion, even at temperatures of 2000 °C. Another similarity between these two metals is their extraterrestrial origin. More precisely, there is very little iridium on our planet, therefore it is found in high concentrations in places where meteorites fall.
Iridium (Ir)
Iridium is a fairly young metal in the scientific world, as it was discovered in 1803 by the chemist Smithson Tennant. He treated platinum with a mixture of nitric and hydrochloric acids, which has a very unusual name - aqua regia. And as is clear from the composition of this solution, after using it you will not become a “drunken master”, like Jackie Chan in the film of the same name, but rather a dead master. For the word “vodka” originally meant plain water and only after the 14th century began to be used to mean an alcoholic drink.
With the help of aqua regia, Mr. Tennant was able to obtain in pure form those impurities that were in platinum, namely osmium and iridium.
As mentioned earlier, there is very little iridium - approximately 3 tons of this metal are mined per year. For comparison, silver production, according to some sources, exceeds 27,000 tons per year.
Basis of the study
The study is based on a method of treating oncological diseases (and some others as well) that is already used in practice - photodynamic therapy (PDT). The main heroes of this method are photosensitizers and light.
Photosensitizers*
are substances that increase the sensitivity to light in biological tissues.
Sensitizers are quite picky, that is, they accumulate only in those tissues that need to be changed for the further procedure of light irradiation.
When light penetrates the target tissue, a photochemical reaction occurs - molecular triplet oxygen (3O2) is converted to singlet oxygen. In addition, highly active radicals are formed. Together, this leads to the death of cancer cells.
Scientists cite the example of photofrin and aminolevulinic acid as the most common photosensitizers in PDT therapy. However, in recent years, metals with high luminescence coefficients have received increasing attention because they have unusual and useful photochemical and photophysical properties. For example, TLD1433 (ruthenium) for PDT treatment of the bladder and WST11 (palladium) for the treatment of blood vessels.
Research results
So why not use iridium, the scientists thought. But first you need a mechanism that will allow you to use this metal. The patient will not take iridium orally, like regular tablets. HSA) comes into play
), which, due to its properties and quantity (about 55% of all blood proteins), is an excellent carrier of various substances (in our case, drugs). Simply put, HSA can be used to deliver anti-cancer drugs to the desired area of the patient's body, as has already been demonstrated in previous studies using osmium, ruthenium and palladium.
Image #1
In the study we are reviewing, scientists created a maleimide-functionalized octahedral organo-iridium(III) complex Ir1 ( 1a
) in combination with HSA. This complex (Ir1-HSA) turned out to be significantly more effective in increasing phosphorescence compared to “pure” Ir1, that is, without HSA.
In the dark, Ir1-HSA is largely nontoxic to normal cells, but exhibits strong photo-cytotoxicity towards cancer cells and their spheroids (cellular formations).
The synthesized Ir1 was stable for 12 hours in the dark and after 1 hour of blue light irradiation. In this case, it was necessary to check the carbon-carbon bond (C=C). To do this, the reaction of the complex of Ir1 and cysteine (Cys) was carried out in a molar ratio of Cys: Ir1 - 2:1 in [D6]DMSO/D2O at a temperature of 298 K for 30 minutes. As a result of proton magnetic resonance, scientists found the peak value of vinyl protons of maleimide groups at around 6.62 ppm (parts per million). When cysteine was added, the peaks disappeared, but then reappeared in the range of 2.9...3.9 ppm. Scientists attribute this to cysteine conjugation.
Next, the scientists tested whether the free thiol Cys34 from HSA was able to react with C=C. To do this, 30 μM (micromole) Ir1 was incubated with HSA (0-120 μM) for 1 hour. Next, the resulting reaction products were separated using reverse-phase high-performance liquid chromatography (RP-HPLC).
When the HSA reached 120 μM, the Ir1 peak disappeared completely (HRS:Ir1 ratio = 4:1). Thus, the thiol (SH) content was 0.27±0.1 mol SH per 1 mol of HSA ( 1c
). Therefore, the concentration of free SH groups from 120 μM HSA is 32.4 ± 1.2 μM. At this indicator, a reaction occurs with 30 μM Ir1, leading to the formation of an adduct (direct connection of molecules) of Ir1 and HSA in a 1:1 ratio.
Pure Ir1 did not show strong emission in aqueous solution, unlike the Ir1-HSA complex ( 1d
).
The higher the HSA concentration, the stronger the phosphorescence of Ir1 itself became ( 1e
).
Image #2
In order to remove free thiol groups from HSA, 100 μM cystine was added to the solution for 24 hours at a temperature of 277 K. The resulting product was combined with Ir1 for 30 minutes. Observations showed a significant decrease in phosphorescence. In the case of the HSA-Cys34 and Ir1 conjugate ( 2a
) the situation was the opposite, and this suggests that it is the free thiol Cys34 that is the connecting link (more correctly, the binding domain) for Ir1.
Now it was necessary to study HSA in more detail, breaking it down into its components. Human serum albumin has one chain of 585 amino acid residues, among which scientists needed to find exactly those that enhance Ir1 luminescence. For this purpose, a luminescent analysis of the interaction of Ir1 with various amino acids was carried out ( 2b
and
2c
).
And as we can see in graph 2b
, the leader among amino acids by a huge margin was histidine (His), which enhances the luminescence of Ir1 by 37 times.
Having understood a little about what and how it works inside the constituent components of the Ir1-HSA complex, scientists moved on to practical application, that is, to experiments.
First, 0.4 millimol Ir1 was diluted in 20 ml MeOH:H2O, 0.4 millimol HSA was added and stirred for 1 hour. Next, the distribution of Ir1-HSA in living lung cancer cells (A549) was examined using confocal microscopy.
Already after 30 minutes, Ir1-HSA was largely concentrated in the cytoplasm of cancer cells. At 60-120 minutes from the start of incubation, the complex penetrated into the nuclei of cancer cells.
Image #3: Confocal microscopy of A549 lung cancer cells.
However, it is worth noting that not the entire complex penetrated into the nuclei of cancer cells, but only Ir1. Immunofluorescence testing showed that HSA is simply absent in the nuclei of cells exposed to Ir1.
Image No. 4: Immunofluorescent analysis of the presence of HSA in lung cancer cells.
But HSA does not disappear without a trace, it simply remains in the cytoplasm and in the membrane of the cancer cell nucleus. It turns out that HSA fully fulfills its function: it delivered Ir to the cell nucleus, while it itself remained outside.
Image No. 5: quantum yield and phosphorescence lifetime of Ir1 and the Ir1-HSA complex.
The scientists also tested the quantum yield (exaggerated, strength) and phosphorescence lifetime of Ir1 (by itself) and the Ir1-HSA complex.
The quantum yield of Ir1 was very small (only 0.001), and the lifetime at a temperature of 298 K was 182.7 nanoseconds ( 5a
). But for Ir1-HSA, the quantum yield was already 0.036, and the lifetime was 871.8 ns. This duration of phosphorescence is excellent for the generation of singlet oxygen (1 O2).
Electron paramagnetic resonance spectroscopy using 2,2,6,6-tetramethylpiperidine as a spin trap revealed 1O2 generation in pure Ir1 and in the Ir1-HSA complex at 465 nm irradiation for 20 minutes ( 5b
). As expected, the 1O2 quantum yield of Ir1-HSA was significantly higher (0.83) than that of Ir1 (0.06).
It was also necessary to check the extent of the effects of Ir1-HSA and Ir1 on cancer cells and on healthy ones. Three variants emerged as cancer cells: lung cancer A549, hepatoma Hep-G2 and cisplatin-resistant lung cancer A549R. MRC-5 (lung) and LO2 (liver) were used as healthy cells. The experiment was carried out in two lighting variations: complete darkness throughout the entire experiment and blue light.
Cells were incubated with Ir1 or Ir1-HSA for 2 hours, washed with sodium perborate and irradiated with blue light for 20 minutes or left in the dark (second version of the experiment). After this, the cells recovered within 46 hours.
The effect of Ir1 on A549 cells both in the dark (89.6 μM) and under light (53.3 μM) was practically absent.
Table of effects of Ir1 and Ir1-HSA on cancer cells: the higher the number, the less impact (i.e. more cancer cells remained unharmed).
But Ir1-HSA showed completely different results. In the dark, the tested complex had no effect on cancer cells, but when illuminated, the degree of its cytotoxicity increased significantly. Ir1 and Ir1-HSA showed similar results, as we can see from the table above, in relation to other cancer cells. At the same time, healthy cells both in the dark and under light were not affected by Ir1 and Ir1-HSA.
Finally, the scientists analyzed reactive oxygen species (ROS) inside cells after light irradiation. In the dark, as expected, no ROS were detected. But in cells that were exposed to light irradiation after application of Ir1-HSA, ROS were detected ( 5c
).
For a more detailed introduction to the study, I strongly recommend that you look at the scientists’ report and additional materials to it.
Epilogue
With this study, scientists did not try to reinvent the wheel in the form of the use of rare metals in photodynamic therapy, because this had already been done previously with osmium and palladium. However, no one had ever tried using iridium, which the researchers decided to fix. Their work was not useless, since iridium showed excellent results in the fight against cancer cells of various types, without affecting healthy ones.
Cancer is one of the most common diseases, claiming millions of lives every year. The invention of new methods of combating this disease and the improvement of existing ones must and will continue. Of course, we are still far from a total victory over cancer, but scientists around the world continue their struggle in laboratories, like millions of patients in the wards.
We should also not forget the factors that lead to the occurrence of cancer.
Some of them (ecology, bad habits, etc.) are within the power of a person to eliminate. Friday offtopic: No one in their right mind would be happy about oncology.
Such diagnoses make you give up and forget about everything. But you can never give up. If you don’t fight, the diagnosis will definitely prevail. And if you fight the disease, there is always a chance to defeat it. So why not use this chance? Thanks for reading, stay curious and have a great weekend guys.
Thank you for staying with us. Do you like our articles? Want to see more interesting materials? Support us by placing an order or recommending to friends, 30% discount for Habr users on a unique analogue of entry-level servers that we invented for you:
The whole truth about VPS (KVM) E5-2650 v4 (6 Cores) 10GB DDR4 240GB SSD 1Gbps from $20 or how to properly share a server? (options available with RAID1 and RAID10, up to 24 cores and up to 40GB DDR4).
VPS (KVM) E5-2650 v4 (6 Cores) 10GB DDR4 240GB SSD 1Gbps free until spring
When paying for a period of six months or more, you can order here.
Is Dell R730xd 2 times cheaper?
Only we have
2 x Intel Dodeca-Core Xeon E5-2650v4 128GB DDR4 6x480GB SSD 1Gbps 100 TV from $249 in the Netherlands and the USA!
Read about How to build a corporate infrastructure. class using Dell R730xd E5-2650 v4 servers costing 9,000 euros for pennies?
Artificial iridium
Iridium, like many other metals, is an extremely rare, but very valuable and widely used metal. The demand for such metals is only growing, but their quantity in nature is decreasing and, to the great regret of mankind, is not being renewed. In this regard, the artificial synthesis of precious metals is gaining momentum in the world. Entire companies and laboratories are being created for this purpose. The most advanced and successful technology for the artificial synthesis of precious metals, which is just beginning to be introduced, is cold nuclear transmutation. This method is recognized throughout the world and today it is used to synthesize artificial platinum. Work is underway on the remaining metals. On the territory of Russia he is engaged in artificial synthesis.
Gold $45 per 1 gram
Gold is the main precious metal, recognized as such throughout the world since ancient times. Gold seems to have been created by nature itself for minting coins and making jewelry: it is found exclusively in its pure form, ductile and resistant to corrosion, homogeneous, compact, in short, an ideal, in a sense, metal. Now you can buy gold in jewelry stores and in banks.
Interesting Facts
Iridium salts are very diverse in color. Thus, depending on the number of added chlorine atoms, the compound may have a copper-red, dark green, olive or brown color. Iridium difluoride is yellow in color. Compounds with ozone and bromine are blue in color. Pure iridium has very high corrosion resistance even when heated to 2000 degrees.
In rocks of terrestrial origin, the concentration of iridium compounds is very low . It seriously increases only in rocks of meteorite origin. This criterion allows researchers to establish important facts about various geological structures. Only a few tons of iridium are produced on earth.
The Young's modulus (also known as the longitudinal elastic modulus) of this metal is in second place among known substances (only graphene has more).
Sources
- https://chem.ru/iridij.html
- https://mysamocvet.ru/metally/iridij/
- https://ProDragmetally.ru/dragotsennye-metally/iridij.html
- https://himsnab-spb.ru/article/ps/ir/
- https://metallsmaster.ru/iridij/
- https://k-tree.ru/tools/chemistry/periodic.php?element=Ir
- https://TheMineral.ru/metally/iridij
- https://vplate.ru/metally-i-splavy/vse-ob-iridii/
- https://jgems.ru/metally/iridij
- https://calcsbox.com/post/iridium.html