The gold alloy contains a certain proportion of this precious metal, the rest being alloys. This variety differs from pure gold in its main properties. In most cases, you can also identify a fake by its external characteristics. A mixture of ligatures and precious metal was widely used in the past and has not lost its popularity today. Modern gold alloys are represented by a large number of types. You can find out how they differ by studying the tables containing the percentages of ligatures.
Pure gold and its characteristics
It is rarely found in its pure form. Mostly it is native gold. There are natural formations that are characterized by large size. Each has its own name, because it is a rare occurrence. But in this form the metal is not used in jewelry, the space industry (when processing satellites), orthopedic dentistry, or industry. The reason is high softness, but strength, on the contrary, is low.
The precious metal is often used to form gold and foreign exchange reserves.
Pure gold
Pure gold is found in the periodic table under atomic number 79. It has the following characteristics:
- atomic mass – 196.966569 g/mol;
- metal density – does not exceed 19.32 g/cm3;
- melting point – 1064.18 °C;
- the precious metal boils at a temperature of 2856°C;
- thermal conductivity – 318 W/m*K.
Au is hard and becomes liquid when combined with mercury (forms an amalgam).
Other features of the precious metal
Gold is one of the heaviest metals known. Its density is 19.3 g/cm3. An ingot weighing 1 kilogram has very small dimensions, 8x4x1.8 centimeters. This is the standard size of a bank gold bar of this weight. It is comparable to the size of a regular credit card, although the bar is slightly thicker.
Only a few elements are heavier than gold: plutonium, osmium, iridium, platinum and rhenium. But their content in the earth’s crust, even taken together, is much less than this precious metal. Moreover, plutonium (chemical symbol Pu, not to be confused with Pt - this is the symbol of platinum) is a radioactive element.
The chemical composition of gold provides its physical properties. So, the main properties of this metal that make it unique include:
- Malleability, plasticity, ductility. It is very easy to flatten or stretch. So, from just one gram of gold you can get a wire 3 kilometers long, and the area of thin sheets obtained from 1 kilogram will be 530 square meters. Ultra-thin sheets of gold foil are called “gold leaf.” They cover, for example, church domes and the interior decoration of palaces. Thanks to its plasticity, a small amount of yellow metal can cover gigantic areas.
- Softness. High-grade gold is so soft that it can be easily scratched even with a fingernail. This is why bullion in jars is sold in sealed plastic packaging. If even one small scratch is noticed on it, it will be considered defective. In order to make gold more durable, other metals are added to it when making products. This property has ensured the high popularity of the king of metals in the jewelry industry.
- High electrical conductivity. Due to this chemical property, gold is highly valued in electrical engineering and industry. Only silver and copper conduct electricity better than it. At the same time, gold hardly heats up: in terms of thermal conductivity, diamond, silver and copper are higher than it. Together with its property of oxidation resistance, gold is an ideal substance for the manufacture of semiconductors.
- Reflection of infrared light. The finest gold plating applied to the glass does not transmit infrared radiation, leaving the visible part of the spectrum. This property is actively used in astronautics when it is necessary to protect the eyes of astronauts from the harmful effects of the sun. Spraying is often used in the mirror system of high-rise buildings to reduce the cost of cooling rooms.
- Resistant to corrosion and oxidation. Ingots that are stored in accordance with the rules are practically not subject to any chemical influence even when exposed to air. So the greater preservation of gold ensured its high popularity.
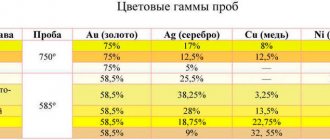
Gold samples in various alloys
A hallmark is a type of mark that determines the weight content of Au in the precious metal. Today in Russia the metric system is used for this. Based on it, according to GOST 30649-99, alloys with special properties and different colors are obtained. The most common:
- 999 or 99.9% - pure gold, the content of precious metal is the highest (but a small percentage of impurities is still acceptable), even fewer additives can only be in a nugget;
- 750 or 75% is a more common option, the amount of Au is significant, which increases the value of jewelry and other products, but this reduces strength;
- 585 or 58.5% is the most popular type of yellow precious metal; it is distinguished by the best ratio of alloys and gold, which allows one to obtain excellent characteristics (high wear resistance, strength, moderate ductility are noted);
- 500 or 50% - the gold content is at the lower limit, which allows the alloy to be classified as precious metals, the strength is higher than that of products of the 585 grade, but the value is lower, which affects the price (products are more affordable);
- 375 or 37.5% is a rarer alloy, but is still used, the Au content is minimal, the value is low, however, jewelry of this type is resistant to mechanical damage, although the price is closer to premium jewelry.
Note: The main types of alloy: copper, silver, platinum group substances, brass and other precious and base metals.
999 standard is the highest standard of gold
Application
The properties of the precious metal (corrosion resistance, ductility, malleability) have found a place for it in human life:
- Industry, mainly electronics. Tablets, phones, computers cannot be made without precious metals. This industry sector consumes more and more precious metals every year.
- Dentistry. Dentures and crowns are still in demand. Although consumption here is declining, more modern and reliable materials have appeared.
- Pharmacology. Preparations containing gold are used in the treatment of tuberculosis, lupus erythematosus, and rheumatoid arthritis. Metal isotopes began to be used in the treatment of oncology. Research shows that radioactive gold may help treat AIDS.
- Food industry. Those who want to try coffee, beer, or chocolate with gold flakes can already do so. Gold is officially registered as a food additive. Remember, don’t confuse it - this is E175.
- Jewelry absorbs the lion's share of the yellow metal's production.
Gold ring with sital
Interesting: the British Medical Journal published the results of a study of the remains of Diana de Poitiers, the favorite of King Henry II of France. At 60, she looked like she was 30. The reason (according to the researchers’ conclusion) is the amount of gold contained in the bones and hair of the favorite’s remains. It exceeded the norm tens of times. Diana took elixirs with precious metal, which were brought to her from Egypt.
Alloy metals in alloys with gold and their properties
In ancient times, purer gold was often used, but the size of the coins quickly changed with intensive use (they were worn out), so they began to use a different technique for producing the precious metal. Alloys were added and proportions were selected that would make it possible to create a material with the most suitable characteristics: high value and strength. The properties of the additives were taken into account. List of the most common ones:
- silver for softness: the main advantage of introducing such an alloy is the ability to increase malleability, ductility, in addition, the precious metal acquires a lighter shade, when choosing the right ratio it becomes white, but the more silver, the lower the value of the product;
- copper for a red tint: used for the purpose of alloying with gold, allows you to change the Au strength indicator (products better withstand mechanical loads), as well as its color - the precious metal acquires a red tint, unlike silver, by adding a small proportion of copper (14-15%) will make it red;
- nickel for hardness: it is characterized by increased hardness and resistance to corrosion, which allows the precious metal to be used in dentistry and the jewelry industry, the disadvantage is the risk of developing allergies when products come into contact with the skin;
- palladium for refractoriness: to obtain a material of higher hardness, the chemical formula changes - palladium is added, at the same time the cost of the products increases, since this alloy also belongs to precious metals, and also contributes to a change in color to a lighter (white);
- platinum for whiteness: used to change the color of the alloy; it is enough to increase the platinum content to 8.4% to obtain a white precious metal; in addition, the addition of such an alloy contributes to an increase in the elasticity of gold and the melting point, which somewhat complicates the work with products;
- ruthenium for strength: increases the melting point, as a result, to change the shape of the product you need to heat it more, gold becomes stronger, but ruthenium does not affect its shade;
- iridium for wear resistance: it is not used to produce an alloy of a different color, but is used to change the properties of the precious metal, as a result, gold becomes even more elastic than when combined with iridium, products are much less amenable to processing;
- osmium for elasticity: the alloy in terms of the degree of impact has many properties in common with ruthenium, iridium, when such a metal is introduced into the composition, the melting temperature changes - it increases, working with the material becomes more difficult, but elasticity increases, its resistance to deformation, but will not withstand a pinpoint impact ;
- cadmium for fusibility: unlike osmium, iridium and other alloys, cadmium, on the contrary, reduces the melting point of the gold mixture, which makes it easier to work with; the precious metal becomes more ductile, but at the same time fragile, since it does not always withstand mechanical loads exerted during the operation of the product, in addition, the gold alloy changes color slightly - a greenish tint appears, but there are other alloys to obtain such a tone, cadmium more often performs other tasks;
- zinc for a green tint: its properties are similar to cadmium, it also contributes to the appearance of a green color, but by choosing a suitable base (ligature), this disadvantage can be eliminated; in addition, zinc increases the hardness of the material, but at the same time it is easier to damage it, because it increases degree of fragility, with pinpoint impacts the product can be destroyed, in addition, the ligature helps to reduce the melting point;
- aluminum for plasticity and unusual shades of gold alloys: one of the types of alloys used in the production of various precious metals, aluminum is introduced into the composition of the violet-colored alloy.
Interesting: A superhard metal based on Au and Ti (titanium) was recently invented.
Precious metals and impurities
Manufacturing
The technology for producing a gold alloy implies some mandatory conditions that are absolutely necessary to adhere to:
- Precious metals are the first to be melted, as they are prone to react with oxygen.
- When it comes to a combination of silver, gold and copper, the latter metal is added as the final metal due to its heat capacity. If this is not observed, the resulting mixture may harden unevenly.
- It is worth considering that the alloy used for joining has a higher melting point, unlike noble metals.
To determine the amount of a particular metal in a product, a special formula is used. It means that 1000 divided by the sample will be equal to the mass of the alloy in grams, divided by the presence of the mass of gold in the product.
The principle of alloying gold with metals
More suitable for processing is a material that is characterized by ductility, low melting point, and strength. The choice of ligatures and their combination with each other allows you to find the best option, taking into account the given characteristics. When one of the additives is introduced, the composition of the material changes. To correct the result obtained, another additive is introduced. For this reason, the metal may contain 1-2 alloys or more.
Note: The homoizing treatment procedure is carried out in order to increase the degree of homogeneity of the precious metal, the desired result is achieved by reheating to a temperature of +700°C, further holding the alloy improves the structure.
Effect of impurities on the melting point of gold
The melting point of gold is important for jewelers. When making jewelry, craftsmen try to use more fusible impurities. After all, if gold melts for too long, it slows down and complicates the work of the master.
The melting point of the most popular 585-carat gold ranges from 880°C to 1280°C. The melting point closest to this indicator is copper (1083°C) and silver (962°C). But palladium is a more refractory element with a coefficient of 1554°C.
The appearance of ready-made ligatures greatly simplified the process of creating jewelry. They already contain all the elements in the required proportions to obtain a certain sample and color. Some give it the necessary plasticity for handwork, rolling and drawing gold wire, while others give it fluidity (for precision casting using lost-wax openwork models).
Alloys of various colors of gold
The choice of alloys is made taking into account the properties that they exhibit when combined with Au. At the same time, they do not focus on the characteristics of the metal itself, which acts as an additive. The result of combining precious and non-precious materials:
- yellow is a classic combination of gold, and in addition also copper and silver: the classic version, to obtain a yellow metal, gold must exceed other additives in quantity, a special, more saturated color is obtained thanks to the combination of silver and copper, and it is the latter of the alloys that enhances the depth of color , and silver does not allow increasing the intensity of the red tone;
- red is a popular duet of Au and Cu: a metal of this color began to be used in Russia in the 18th century, a reduced copper content and additional additives (silver) make it possible to obtain rose gold, it is based on a red alloy, but the properties are different, because the ratio components are significantly different;
- white - adding various alloys for an exquisite white color: the metal has a different name - “white gold”, but the main component is still Au, the platinum group metals used as alloys, as well as manganese, nickel, change its classic yellow color, as a result products may have other properties different from those exhibited by platinum jewelry - the fusibility increases, due to which the variety of shapes and designs of jewelry increases;
- black - an alloy as a result of oxidation with the help of cobalt: a ring or bracelet acquires this color if oxidation with cobalt occurred during manufacturing, an alternative option is dressing with the addition of carbon, patination and rhodium plating also imitate a similar color;
- green - the alloy includes gold with a silver alloy: such products have a name - “electrum”, a metal containing a certain proportion of Ag, characterized by a faint green tint, this color is more pronounced if gold is alloyed with cadmium, but such jewelry is not recommended for long wear may cause harm to health;
- blue and blue - a spectacular alloy with indium: not strong enough, so platinum group metals are often added to the composition, indium gives a blue color, and blue gold is an alloy based on iridium;
- purple is a mysterious alloy of gold with aluminum and palladium: a rare combination, therefore it is not found often, this method is mainly used for making inserts, the disadvantage is fragility.
Interesting: in Ancient Greece, the alloy of gold and silver had a specific name - electron.
Gold of different shades
How to recognize a fake?
Since there are now a lot of fakes that look very much like gold, show them to a specialist when purchasing. If it is not there, then look at the sample and carat weight. Although this method will not always help, since they are now testing fake jewelry as well. Inspect the item if you see any wear on the edges and if it is fake, you may notice other metal.
Since ancient times, gold has been tested by tooth: if you bite it lightly, traces will remain on it, but on a fake it will not, unless lead is added to the alloy. If you put gold in a plate with vinegar, the fake will immediately darken. You can also check with iodine: if you drop it on the jewelry, the fake will darken, but the gold jewelry will not change its appearance. There is a special pencil in medicine; if you run it over the product, the fake will darken. Some counterfeits can be detected using a magnet.
In order not to fall for the tricks of scammers, purchase gold from reputable or verified stores. Avoid buying jewelry in passages, stalls, and online stores. Contact professionals to verify the authenticity of the product, its quality and value.
As we can see, many alloys are similar in appearance to gold, which is passed off as real jewelry. When purchasing, be careful not to purchase a fake that has nothing to do with gold. You can purchase a high-quality fake that is indistinguishable from the original. Nowadays, counterfeits that imitate gold are a profitable business for fraudsters and smugglers. Check the products you buy. Happy shopping!
Imitation gold
The metal may not contain Au, but must have similar external characteristics to be considered similar to the original. Such products have no value, but they have a wide range of applications, for example, the production of costume jewelry is the main direction. List of metals that imitate gold alloy:
- bronze: copper with the addition of aluminum, the first component gives a red tint, the second brightens the tone;
- vermeil: silver with gilding, the appearance of Au is preserved, but the products are lighter;
- Hameltonmetal: a method of coating products that was previously often used; the metal contained copper and zinc.
Methods for obtaining colored gold
There are several ways to obtain colored gold:
- Applying a special coating to the surface of the finished product. It is carried out due to the thermal and chemical reaction of metal alloys with the environment.
- Oxidation. This method involves creating decoration with artificial colors. An oxide layer is applied to the surface of the jewelry.
- Patination. In this case, chemical treatment of the alloy, which contains copper, occurs.
- Oxidation. Gold products are oxidized in air for 30-60 minutes at a temperature of 750-950 degrees Celsius. Thus, cobalt is used to obtain black gold.
- Rhodium plating. Application of rhodium plating on finished products.
- Galvanostegy. This method involves the use of solutions with the addition of special dyes that form a special layer on the jewelry and give it the desired color.
- Gold recoloring is the youngest, recently discovered method of obtaining colored gold. At the atomic level, gold is milled with an ion beam. The structure and depth are selected, light waves are controlled. Thanks to the ability of metals to absorb and reflect waves, any shade of gold can be obtained.
Thus, gold has been valued from time immemorial and is still considered a noble metal. A large selection of alloy compositions makes gold products accessible to people with different income levels.
Decoding the markings of alloys that contain gold
Materials are marked for ease of handling. For precious metals, in addition to the letter, an additional numerical designation is generally accepted - fractions of thousandths, and for base alloys - hundredths (%). Common options found in GOST:
- gold – Evil;
- silver - Wed;
- platinum – Pl;
- copper – M;
- zinc – C;
- nickel – N;
- cadmium – Kd;
- palladium – Pd.
Medical gold alloy
Recommendations for care, cleaning and storage
Gold jewelry requires regular care and careful handling. If you follow the basic rules, they will serve for many years:
- It is necessary to store gold jewelry in individual cases and fabric bags. It is better to place chains in elongated boxes so that all the links are straightened and do not break.
- From time to time, jewelry should be washed in warm water and soap. Do not use hard brushes or aggressive chemicals.
- A special paste for cleaning gold jewelry, which can be purchased at a jewelry store, will help you get rid of darkening. It is better to trust the cleaning of antique and especially valuable jewelry to professionals.
- Gold requires careful handling: it is better to remove jewelry during swimming, physical work, and sports.
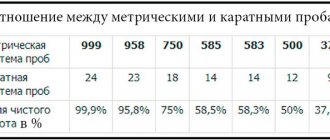
World systems for measuring the purity of gold precious metals
At different times, a wide variety of methods were used to determine the Au content in a metal alloy. At the same time, the number of ligatures that were added at the manufacturing stage varies: from 10-12% to 80%. But the best option in terms of appearance and properties is the one that contains 30-40% additives. There are only 4 known ways to measure the purity of a precious metal:
- widely used metric system: the fraction of the precious metal is determined in thousandths (range from 0 to 1000);
- carat system, applicable in the USA and Europe: the value taken as a basis is 24 carats, this is a precious metal without impurities; accordingly, the gold content in different alloys is determined as the number of parts of 1/24 of the pure substance (corresponding to 1 carat);
- spool system of Tsarist Russia: the basis was taken as a measure - 96 samples of pure precious metal, corresponding to the number of spools in 1 Russian pound;
- lot system of Old Europe: metal of 16 lots was considered pure; the lower this number, the lower the value of the products.
Metal mining from ancient times to the present day
Gold is an amazing metal. All minerals are mined to be used and consumed, but they are mostly hoarded. States and individual citizens.
From Egypt to America
It is believed that the Egyptians were the first to mine the yellow metal. Chronicles recorded metal mining in the Arabian-Nubian province. There were rich deposits there. Even in the time of Thutmose III, about 50 tons of gold were mined there per year. And in total, during its operation, the deposit produced more than 3,500 tons of metal.
However, the deposit has become depleted. Many historians associate the decline of Egypt precisely with the fall in the amount of precious metal in the state.
New deposits have been found in Europe (Spain, Austria-Hungary). And with the Spanish conquests in America, solar metal poured into the Old World in a torrent.
Over the entire history of mankind, about 167,000 tons of the precious metal have been mined.
Of these, 12% were used in industry, and of 50% people made jewelry.
In any case, the extraction of precious metals stimulated the development of metallurgy, and at the same time international trade.
Modern gold mining
The days of the gold rush that shook countries are long gone. Cruel romance, when thousands of people went bankrupt and many died, is a thing of the past. Although only a few of them became obscenely rich.
Now gold mining takes into account profits and overhead costs, and does not take a step without preliminary exploration of deposits, assessment of economic risks, and construction of mines using the proper equipment.
In modern times, gold ore is mined mainly at great depths or from depleted placer deposits. Gold in nature is not a renewable resource, but it has been mined since time immemorial. Even in the old days, the cream was skimmed from the richest deposits, and we, descendants, are left with the hard work of choosing gold from already “refined” places. Or, as an option, the yellow metal is mined at great depths.
We recommend: COBALT - a generous gift from mountain spirits
How alloys can change the appearance of a product
According to GOST, the difference between metals of the same group can be small or significant. However, the gold content in the alloy is always the same, despite the change in the proportions included in the composition of the alloys. The shade changes when the amount of basic additives varies: silver, copper, which determines the depth of color.
Table 1: Alloys with Au in the composition, 375 samples
Metal based on Ag, Au, Cu is presented in the following options:
| Au, % | Ag,% | Cu,% |
| 37,2-37,8 | 1,5-2,5 | 59,5-61,5 |
| 9,5-10,5 | 51,5-53,5 | |
| 15,5-16,5 | 45,6-47,4 |
Gold purity 375
Table 2: Alloys that contain Au 500 grade
| Gold, % | Silver, % | Copper, % |
| 49,7-50,3 | 9,5-10,5 | 39,2-40,8 |
| 19,5-20,5 | 29,2-30,8 |
Table 3: Au 583 alloys
| Gold, % | Silver, % | Copper, % |
| 58,0-58,6 | 7,5-8,5 | 32,9-34,5 |
| 19,5-20,5 | 21,1-22,3 | |
| 29,5-30,5 | 11,2-12,2 |
Table 4: Au 585 based alloys
| Au, % | Ag,% | Cu,% |
| 58,5-59,0 | 7,5-8,5 | 32,5-34,0 |
| 19,5-20,5 | 20,5-22,0 | |
| 29,5-30,5 | 10,5-12,0 |
Hallmark 585
Table 5: 750 grade alloys
| Au, % | Ag,% | Cu,% |
| Au, % | Ag,% | Cu,% |
| 74,7-75,3 | 12,0-13,0 | 12,0-13,0 |
| 14,5-15,5 | 9,5-10,5 |
Table 6: 958 alloys
| Au, % | Ag,% | Cu,% |
| Ag,% | Ag,% | Cu,% |
| 95,5-96,1 | 1,5-2,5 | 1,7-2,5 |
Description of the technique
Each alloy was pre-melted (800g for 18ct alloys, 700g for 14ct alloys, 600g for 9ct and 10ct alloys). Melting was carried out in a graphite crucible in an open furnace with induction heating. The metal was then cast into ingots. An open furnace was used to utilize mechanical mixing for better ingot homogeneity.
The pouring temperature depended on fineness and color, and the alloys were divided into groups according to composition. The metal was protected by purging inert gas into the crucible during melting and pouring. The ingots were then rolled and cut into pieces and remelted in a closed, controlled atmosphere casting machine. After preliminary melting, alloy samples were submitted for DTA analysis*. The pouring temperature was 100°C higher than the liquidus point measured in a closed casting machine.
For each alloy, a maximum of five casting operations were performed in a closed casting machine using pre-molten material. The following samples were obtained for subsequent research:
- 20 x 8mm rectangular ingots for hardening testing and specimen making for density measurements and color determination (and nickel testing). These ingots were cast for all compositions.
- Rectangular ingots 70 x 5 mm for the production of samples for drawing tests. These ingots were cast for malleable alloys only.
- 6 x 6 mm square ingots for wire drawing in tensile tests in forged material. These ingots were cast for malleable alloys only.
- Set of 16 samples for testing hardness and determining the average grain size (24 x 12 mm, 4 mm plates). These samples were cast for all compositions.
- Set of 30 specimens for testing tensile strength in cast alloys.
The closed casting machine consisted of two communicating chambers. The mold and flask were in the lower chamber, and the melting crucible was in the upper chamber. The pouring occurred through a hole in the bottom of the crucible after opening the sealing hole.
The molds were preheated to 320°C in an oven. The molds were sized to fit into a controlled atmosphere casting machine. The preheated mold was placed into the machine after the crucible was filled with metal, but before the machine was closed and the casting cycle began (an automatic program was used). This procedure was preferred in order to cast ingots of all alloys under the same conditions and avoid errors that can occur when casting by hand.
After casting, the chamber with the castings was quickly opened and the ingot was immediately lowered into water.
The mold with the plates was preheated to 600°C, and the container with the tensile test specimens was preheated at 700°C to avoid incomplete filling. In this case, the containers were also placed in the casting machine before closing and starting the casting cycle. After casting, the molds were cooled with water after 6 minutes for alloys containing grain refiners, or after 20 minutes for alloys containing only deoxidizers.
An ingot 20 mm wide and 8 mm thick was rolled in several rolling passes to determine the hardening curve of the alloy. Hardness was measured on cast metal (8 mm thick) after 40% rolling (5 mm thick), after 60% rolling (3 mm thick), after 80% rolling (1.5 mm thick) and after 90% (thickness 0.8 mm). Ten samples measuring 16 x 8 mm were cut from a sheet of 1.5 mm thickness. Of these, 5 samples were tested for density, 2 were used to determine color, and 3 were used to measure hardness after annealing.
The hardening curve was determined only for ductile alloys. Alloys for investment casting were rolled from a thickness of 8 mm to 1.5 mm, then the samples were cut.
Alloys containing nickel were rolled to a sheet 2 mm thick. The sheet was cut into two parts, one of them was annealed. The thickness was then reduced to 0.5 mm and three 12 mm diameter specimens were cut (with a 1 mm diameter hole) for the nickel migration** test according to the UNI EN 1811 standard.
A 70 mm, 5 mm thick ingot was rolled to a thickness of 2 mm and then annealed. After rolling the sheet to 0.4 mm, five samples with a diameter of 70 x 70 mm were cut. These samples were annealed under suitable conditions (see Table 1) and were used for deep drawing tests.
A 6 x 6 mm square ingot was rolled to a 1 x 1 mm square section for tensile testing with the following rolling schedule. Rolling from 6 x 6 to 4 x 4 mm square (55% reduction) - annealing - rolling from 4 x 4 - 2 x 2 mm square (75% reduction) - annealing - rolling from 2 x 2 mm to 1 x 1 square section - final annealing. For tensile tests, 1 x 1 mm wire was used. Tensile tests on wire are carried out only in the case of ductile alloys.
Annealing parameters depending on the composition of the alloy are presented in Table 1.
Table 1 Annealing parameters
| Trial | Temperature, оС | Time, min | Cooling |
| 9, 10, 14 ct white | 725 | 18 | quick in oil |
| 18k white | 700 | 18 | quick in oil |
| 9, 10, 14 ct color | 675 | 17 | fast in water |
| 18k color | 650 | 17 | fast in water |
In all cases, annealing was carried out in a static laboratory furnace in a reducing atmosphere.
A set of plates 24 × 12 × 4 mm was made to determine the hardness of alloys after casting or after various quenching methods: 1 plate after casting, 1 after heat treatment, 9 after exposure at different temperatures and for different times.
Heat treatment parameters (temperature, time) depending on the composition are presented in Table 2.
Table 2 Heat treatment parameters
| Trial | Temperature, оС | Time, min | Cooling |
| 9, 10, 14 ct white | 750 | 30 | quick in oil |
| 18k white | 725 | 30 | quick in oil |
| 9, 10, 14 ct color | 700 | 30 | fast in water |
| 18k color | 675 | 30 | fast in water |
Strengthening after heat treatment increased for all alloys (regardless of composition) at 250, 300, 350 °C. The oven holding times were 60, 120 and 180 minutes for each temperature. Therefore, we had 9 hardened samples. All samples were subjected to heat treatment before hardening, according to Table 2.
The hardening characteristics were tested on undeformed cast alloys after appropriate heat treatment. This procedure is simpler and faster, and even if it does not achieve high hardening, it allows the accurate identification of the alloys being hardened. In any case, the data for samples subjected to rapid hardening are comparable, and a classification of alloys in order of increasing hardening can be obtained.
The tensile test was carried out only for cast alloys. The tested samples had a diameter of 2 mm and a length of 12 mm (L0), Fig. 1.
The collected data will be presented below. We will not go into detail here as each test follows a European or ASTM standard.
DTA test
Differential thermal analysis (DTA) allows one to find the solidus and liquidus temperatures (i.e., melting range) of alloys. 25 mg of compressed sample was used. The DTA test was carried out on pre-molten metal to find the pouring temperature in a closed machine. The data was confirmed on the same sample after remelting (the alloys are more homogeneous after remelting).
Density measurement
The density of the alloys was measured according to Archimedes' law, which states that a body immersed in a liquid experiences a buoyant force equal to the weight of the liquid (or gas) displaced by this body.
The following formula was used to calculate the density:
d = w(a)*d(fl)
Where:
d = density of solid sample d (fl) = density of liquid
w(a) = mass of solid sample in air
w (fl) = mass of solid sample in liquid
To measure density, hardened plates measuring 16 x 8 mm and thickness 1.5 mm (5 samples) were used.
Extrusion test
Ductility was assessed by measuring the displacement of the punch when the sample (70 x 70 mm sheet with a thickness of 0.4 mm) begins to break. This information is very important for the production of "stamps".
The plasticity value corresponds to the height of the cup (in mm) pressed into the sheet by the punch. The punch is made of a steel sphere with a diameter of 20 mm and is gradually pressed onto the sheet until the operator detects the initiation of a crack. The sheet is secured with a clamping device to avoid slipping during testing. This is practically the Eriksen method, see ASTM E 643-84.
Tensile and tear test
The tensile test measures ultimate tensile stress (UTS), yield strength (YS) and percentage elongation to break (E%). The first two parameters assess the mechanical strength, and the third - the ductility of the material.
UTS is defined as the ratio between the maximum applied load and the cross-sectional area of the tensile specimen before loading. This is a characteristic property of the material. More specifically, UTS is the maximum uniaxial stress that a material can endure before failure. The yield stress is the stress corresponding to the onset of plastic deformation, that is, the transition from elastic reversible deformation to irreversible plastic deformation. In this work, a 0.1% level of strain strength (YS0.1) was considered.
Strain to break, commonly referred to as percentage elongation, is denoted E% and is measured as the increase in the length of the specimen being broken, expressed as a percentage of the original length of the test specimen.
The tensile test was carried out on two different types of specimens, depending on the type of alloy, i.e. cast alloys and alloys designed for mechanical deformation.
In the case of cast alloys, tensile specimens were obtained directly from the mold. Therefore, these tensile specimens must be accurately tested to keep the number of erroneous tests to a minimum: i.e., the specimens must be free of visible defects and perfectly centered. In a tensile test, if rupture occurs outside the length L0 (Figure 1), the result is discarded since it is believed that stress concentration near the change in cross-section may change the test result. In order to obtain a reliable result, the results of at least 15 tensile specimens were considered: the final result is the average of these tests. In addition, the clamping device was carefully designed to minimize clearance and sample positioning error.
In the case of alloys intended for processing by plastic deformation methods, a square cross-section of 1 x 1 mm wire with a length of 200 mm was used. The samples were annealed after rolling and carefully checked to avoid visible surface defects. A pneumatic clamping device with partial wire wrapping was used. The wire break must occur between the terminals to have the correct result. In this case, a minimum of 15 correct trials were also averaged. The test was carried out according to ASTM E8-00 standard.
In the case of alloys intended for both casting and plastic deformation, tensile tests were carried out using both methods described above.
Hardness Determination
The hardness of materials was measured under various conditions: casting, quenching, hot rolling, hardened alloy. The Vickers method was used with a load of 500 g for 15 s. See ASTM E 92-82 and E 384-99. More precisely, the hardness was measured on samples treated as follows:
1. Directly after casting and after 40%, 60%, 80% and 90% rolling. These measurements were carried out only on alloys intended for plastic deformation.
2. Annealing: 80% rolling followed by annealing in accordance with the parameters of table 1.
3. Heat treatment: samples of cast plates, after hot rolling and hardened samples under 9 different conditions (temperature and time).
Grain size measurement
The average grain size was measured according to the Jeffreys method, that is, by counting the number of grains present in a given area. The cast samples were carefully polished and metallographically etched with a cyanide solution to make the grain visible. See ASTM E 112-96.
Grain size was measured only on cast specimens and not on annealed ones, for both cast alloys and duct alloys.
Color rating
CIELab*** color coordinates were determined from reflectances measured on samples specially prepared for this purpose (hardened and polished 16 x 8 mm plates).
Reflectance spectra of samples to obtain CIELab color coordinates (L*, a* and b*). processed by computer (see ASTM E 308-99) One point in three-dimensional color space is identified for each alloy composition.
Measuring Nickel Release Rate
This test is designed to simulate the rate of nickel release from products that come into direct and prolonged contact with human skin. The UNI EN 1811 standard has been carefully followed. The geometry of the samples of various alloys corresponded to the provisions of the above standard. At the end of the test period, the concentration of dissolved nickel in the solution was determined using inductively coupled plasma spectroscopy (ICP-OES). The nickel release rate was measured in mg/cm2/week and the corrected value was taken into account.
The rate of nickel release was measured only on nickel-containing alloys, that is, on white gold.
Jeweler's comment
Molokanov N.M.
Jeweler, 26 years of experience in jewelry production.
The content of the alloy makes the metal different from others. Today, the composition is often changed, according to GOST, as a result of a decrease/increase in the proportions of gold, copper, silver or other metals, which can affect strength, density, fusibility and, of course, color. Thanks to this feature, the shade of precious metals serves as one of the signs of authenticity.
Where can I buy or sell?
The safest place to buy jewelry is in jewelry stores. Products must have a certificate and passport. You can buy and sell gold in other places:
- in the online store;
- in a pawnshop;
- on Avito and similar resources with private advertisements;
- On the market.
In all these cases, the buyer has no guarantee of quality and cannot return the product if a defect is discovered. However, you can find antiques and jewelry here at lower prices.
Tips for choosing gold jewelry
A few tips will help you make a successful purchase in a jewelry store:
- It is necessary to study the certificate or passport of the product.
- Carefully check the appearance of the product; for this you can ask the seller for a magnifying glass. The surface should be free of scratches, abrasions, and poor-quality polishing. All fastenings and inserts must be strong. In Russia, the return of jewelry is possible only if an undeclared defect is discovered, so you should carefully choose the size of the product.
- You should not buy jewelry to invest money. Jewelry is very difficult to sell at market price. Pawnshops accept them for the price of scrap gold.
- You should trust official documents; popular advice on determining the quality of a precious metal can be misleading. Even a specialist will not be able to correctly assess the quality of gold only by how the product looks.