Due to their high chemical inertness, they do not oxidize or corrode, thereby maintaining their characteristic shine and visual appeal.
Historically, precious metals have been used to make jewelry, and the high cost has placed gold and platinum in the same price range as precious stones.
In our age of high technology, precious metals are widely used in the production of products for the electronic, radio engineering and space industries, in the manufacture of laser technology and medical equipment, household appliances, computers and various gadgets.
Many parts included in hi-tech equipment contain precious metals in microscopic quantities.
Modern technologies for extracting this valuable material from secondary raw materials have turned the recycling of precious metals into a highly efficient production, surpassing in its profitability the extraction of gold, silver and platinum group elements from primary natural raw materials.
List of precious metals
The list of precious metals is presented in GOST R 52793-2007 “Precious metals. Terms and definitions" and legalized by the Federal Law of March 26, 1998. for No. 41-FZ “On Precious Metals and Precious Stones” (Article 1).
According to GOST R 52793-2007 (section 2), only 8 are considered precious metals, here is a list of them:
- gold;
- silver;
- platinum;
- palladium;
- iridium;
- rhodium;
- osmium;
- ruthenium.
As you can see, the list also includes silver, this will be interesting to know for those who are wondering whether silver is a precious metal.
What it is?
Noble metals are distinguished from other elements by their ability to maintain their molecular structure under the influence of an unfavorable environment.
They cannot be decomposed, and the melting point of these elements is extremely high. They do not react with oxygen and do not produce oxides. Alloys with precious metals can only be obtained through complex chemical manipulations using the strongest reagents. The mass share of precious metals in the total production of natural resources is very small, which explains the high price of these elements, as well as their special status.
A complete list of names of precious metals was prescribed in 1998 in the Federal Law “On Precious Metals and Precious Stones”. According to Russian legislation, platinum and five of its group metals, as well as the popular silver and gold, are considered precious.
Areas of use
By nature, gold and other precious metals are endowed with unique consumer qualities , for which gold products or silver crafts are so dear to their owners.
It’s not for nothing that the term “precious” is interpreted in two ways in Russian dictionaries - both “valuable, expensive” and “dear, dear, very necessary.”
Nowadays, the first place among the special properties of precious metals, which are absent in other materials, is their high resistance to the destructive influences of the environment in which they are used.
Therefore, already from the middle of the 19th century, platinum, gold, silver, iridium and other noble elements became the subject of industrial use .
Each material has its own specific technological niche, in accordance with which its certain market value has been established, taking into account the need for it and the complexity of its extraction from natural raw materials.
Below is a summary of the use of precious metals and approximate prices in rubles/gram:
- Gold is used for the manufacture of jewelry and dental prostheses, as a solder or welding material in the manufacture of thermocouples, as an electroplating coating for circuit boards, contacts, and connectors in the manufacture of products for all industries. With the development of nanotechnology, telecommunications, aerospace and nuclear energy, the scope of gold has expanded significantly. Gold is present in targets for nuclear research, used in special casings for neutron bombs, etc. The price of gold is set at 2570.98 rubles/gram.
- Silver is used in contacts of electrical products, in copper-silver solders, in various alloys, and is still used in photography. A large amount of silver is used for the manufacture of high-energy density batteries (silver-zinc and silver-cadmium) and as additives to lead for current leads of lead-acid batteries. Silver-coated mirrors are superior in reflective qualities to their aluminum-coated counterparts. The price of silver is set at 30.41 rubles/gram.
- Platinum still competes with gold in the production of jewelry, is indispensable for the manufacture of heat-resistant chemical laboratory glassware, and is popular in metallurgy as an alloying additive. Electroplating, heating elements, resistance thermometers, relay contacts - platinum is present everywhere. The cost of platinum is set at 1,756.73 rubles/gram.
- Rhodium is used for finishing jewelry, in the manufacture of thermocouples and neutron detectors for nuclear technology. Its heat-resistant properties are used in dies for glass filaments and thermocouples, and chemical inertness is taken into account in the production technology of nitric acid. The cost of rhodium is 5406.96 rubles/gram.
- Palladium, osmium, iridium and ruthenium are successfully used as catalysts for various technological processes in the chemical industry, in the manufacture of measuring sensors, for applying special coatings in products of the defense and space industries, and in telecommunication systems. Platinum group metals are indispensable for the manufacture of technological equipment operated in aggressive environments. Corrosion-resistant, heat-resistant alloys of ruthenium with iridium and platinum are materials for dies for fiberglass and viscose. Executive-class fountain pens are very popular, in which the writing “eternal” nibs, in accordance with their name, are soldered with osmium alloys with palladium and platinum.
For platinoids, as the noble elements of the platinum group are often called, the Central Bank of the Russian Federation has set the following prices:
- palladium – 2220.49 rubles/gram;
- osmium – 854.86 rubles/gram;
- iridium – 3120.22 rubles/gram;
- ruthenium – 559.93 rubles/gram.
You can read more about prices, as well as about methods of donating precious metals, be it metal refined from radio components or scrap jewelry, here.
Application of noble metals in the automotive industry
If you open the car’s operating manual, then in almost every one of them you can find information about which components and in what quantities precious metals are used.
The main metals used in cars are silver, gold, palladium and platinum. Thus, according to the operating manual for LADA 110 cars from 2007, cars of the tenth family use 0.103 grams of palladium, less than 0.1 grams of gold and more than 3 grams of silver. These metals are mainly used in contacts and conductors in the production of relays, breakers, sensors or control units. GAZ cars, for example model 2217, do not use palladium, but about 0.1 grams of platinum and more than 5 grams of silver are used. Even more precious metals are used in the production of KAMAZ trucks.
Natural mining and production
The share of precious metals in the production volume of the global mining industry is only 0.00005%. The content of noble elements in the earth's crust is small; accordingly, the volumes of their extraction are not comparable with the development of other minerals. For example, less than 30 tons of rhodium are mined annually .
However, taking into account the high prices of gold, platinum and other precious metals, the cost of mined precious metals exceeds 5% of the value expression of the volume of production of all minerals.
That is, it is reasonable to estimate the cost of mining precious metals in a proportion that is 100 thousand times higher than the cost of mining any other natural raw material (except diamond).
The extraction of gold and other precious metals plays a special role in the state development system, since the available reserves of precious metals determine the strategic state of the gold and foreign exchange reserves of any country.
With the development of technologies for extracting precious metals from various types of raw materials in industrialized countries, three areas of production have emerged, each of which contributes to the replenishment of state gold and foreign exchange reserves:
- Primary mining associated with the extraction of precious metals from ore and the production of concentrated (enriched) materials, as well as the processing of industrial waste.
- Associated production associated with the extraction of precious metals from polymetallic ores mined for the production of non-ferrous metals.
- Recycling, which is the recovery of primarily gold, silver and platinum from scrap precious metals and waste.
In the Russian Federation, gold production indicators for 2014 were as follows :
- primary production – 232185 kg;
- associated production – 16880 kg;
- secondary production – 38913 kg.
Let's show similar indicators for silver for 2014:
- primary production – 510272 kg;
- associated production – 287978 kg;
- secondary production – 249139 kg.
There are several articles on the topic of independent mining of precious metals on our website:
- Search magnets for gold and silver.
- Metal detector for gold and silver mining.
- How to look for gold with a metal detector.
- We make a metal detector with our own hands.
Where can I buy or sell?
The main lots on the market remain platinum and gold. If you represent a company or have an individual entrepreneur, it is more profitable to buy metals from officially registered brokerage companies that work with leading manufacturing plants.
Expert opinion
Lyudmila Pestereva
Our most experienced gold investor
Ask a Question
It is easier for an individual to cooperate with banks that offer a wide range of services for the purchase of precious metals. For example, buy bullion and put them in your own cell, open a deposit and even make money on your investments.
Which precious metal is the most expensive?
If we are talking about the most popular and popular metals, then the leading place in the price ranking is occupied by palladium, platinum and gold.
However, Californian is rightfully considered the most expensive metal on earth.
It is mined during the operation of powerful nuclear reactors. The price for 1 gram of Californian is 6.5 million dollars. Next on the list of expensive metals is rhodium. It is estimated at 225 thousand dollars per gram.
Designations of precious metals
To designate chemical elements, natural substances and materials of artificial origin, special symbols are used in the form of conventional signs (symbols), codes or abbreviations that provide brief and visual information about the name of a chemical element or similar simple substance.
For noble metals
the following designations are accepted :
- gold - Au (from Latin Aurum);
- silver – Ag (from Latin Argentum);
- platinum – Pt (from Latin Platinum);
- palladium – Pd (from Latin Palladium);
- rhodium – Rh (from Latin Rhodium);
- ruthenium - Ru (from Latin Ruthenium);
- osmium - Os (from Latin Osmium);
- iridium - Ir (from Latin Iridium).
The symbolism of the symbols of precious metals is used for the following purposes :
- when marking jewelry;
- in metallurgy - in labeling information about the chemical composition of non-ferrous metal alloys in which precious metals are present as alloying additives (ligatures);
- when using precious metals as currency values, exchange commodities or other purposes in the financial market;
- when designating coin metals used in numismatics.
When designating precious metals in alloys, Cyrillic letters are used . For example, according to GOST 13498-2010 “Platinum and alloys based on it. Marks" the following designations of precious metals are accepted (clause 3.1):
- Pl – platinum;
- Pd – palladium;
- I – iridium;
- Rd – rhodium;
- Ru – ruthenium.
In particular, the PlRd 93-7 alloy contains platinum and rhodium.
Similarly for gold and silver in GOST 30649-99 “Alloys based on precious metals for jewelry. Brands" the corresponding designations Zl and Sr are adopted (clause 3.1).
For example, the ZlSrPd 585-255-160 alloy contains gold, silver and palladium.
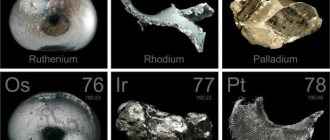
Noble metals from the platinum category
Five more types of precious metals come from the platinum category:
- Palladium (Pd) is the 46th element of the periodic table and has a silvery-white color. The melting point is 1552°C, and its density is 12.02 g/m3. This is the metal with the smallest mass among others from the platinum category. At the same time, it is extremely resistant to aggressive environments and chemicals. Other advantages of palladium include ductility, ease of processing and polishing, and the ability to retain shine.
- Rhodium (Rh) is a white metal with a blue tint, located at position 45 of the periodic table. Among the chemical characteristics, its high density is noted - 12.42 g/m3, as well as its melting point - 1960 ° C. This is one of the refractory metals; along with sufficient hardness, it is also brittle. Rhodium is highly reflective and resistant to water, oxygen, and is not affected by any acids. Only alkaline cyanide mixtures dissolve rhodium.
- Ruthenium (Ru) is an element of the periodic table with atomic number 44. This metal is white in color with a silvery tint and its appearance is very similar to platinum. However, it is distinguished by great hardness with simultaneous fragility, as well as very difficult fusibility. Ruthenium melts at 2950°C, and its density is 12.37 g/m3. A distinctive feature is its resistance to chemical attack. It is the rarest of the platinum group metals.
- Iridium (Ir) is a precious metal located at 77th place in the table of chemical elements, colored white with a gray tint. The main qualities of iridium are high refractoriness, brittleness and at the same time increased hardness. The density is 22.42 g/m3, and the melting point is 2450°C. This is the reason for the difficulties in processing it, since it can only be worked under high pressure and at high temperature. In addition, this metal does not interact with any chemical compounds, be it alkalis, acids or mixtures thereof.
- Osmium (Os) is an element from the group of platinum metals, occupying 76th place in the periodic table. This material is the most difficult to process, extremely brittle, but at the same time it is very hard and extremely refractory. The density of osmium is 22.48 g/m3, and its melting point is the highest among platinum metals - 3047 ° C. A special feature is its pungent odor, as well as absolute resistance to any alkaline or acidic environments.
Product marking
In the production of technical products for various purposes and jewelry, precious metals are used exclusively in the form of alloys with other metals.
Precious metal alloys get their name from their main component (gold alloys, platinum alloys, etc.).
The main materials used for jewelry are gold, silver, platinum and palladium.
Other platinoids (rhodium, osmium, iridium and ruthenium) are added to precious metal alloys in order to adjust the physical and technical properties of the product being manufactured - color changes, ductility and hardness, heat resistance and melting point.
To identify products containing precious metals, markings are used (from German markieren - to put a mark, an iconic mark) with marking information applied in the following ways :
- using a mark (branding) directly on the surface of the product - for jewelry;
- entries in product passports, forms, labels, operating manuals, reference books and catalogs.
Purpose and methods of branding
According to the “Regulations on hallmarking and hallmarking of products made of precious metals...” (Decree of the President of the Russian Federation dated October 2, 1992 No. 1152), all jewelry and household products made from precious metals for sale must be branded with the state hallmark (clause 2 ).
The mark is applied only when the jewelry is made in accordance with all official canons and meets state quality standards.
The absence of a mark clearly indicates that it has not passed the inspection of the state inspection service, or was simply not presented to it and is being sold in circumvention of legal regulations.
Another marking on the product is the name tag, which is an imprint designating the manufacturer who manufactured the product.
The sale of jewelry made of precious metals without a name mark and inspection by the assay service is illegal .
At its core, hallmarking is an assay-technological operation of applying an hallmark to products made of precious metals. The method of its application is determined by the degree of complexity of the shape and purpose of the product on which the marking must be applied.
Currently, three branding methods are used:
- A mechanical or impact method of applying a mark, based on mechanical action on the surface of the jewelry (hitting a tool with a mark on the tip with a hammer after applying the mark to the surface of the product). Until now, about 80% of all products made from precious metals are branded in this way. Only the hammer has been replaced by automated stamping machines.
- Electroerosive or electro-spark method, which is essentially the burning of a mark with an electric spark. This branding technique is convenient for working with fragile hollow products. About 10-20% of all jewelry products are branded using the electrospark method.
- Laser method, in which the outline of the mark is applied with a laser. This mark is small in size, but has clear outlines. It is considered the only way to brand convex and concave hollow products.
How to read the mark?
The mark consists of two marks , which are either applied separately or presented together on one print:
- images of the sample mark;
- identification design - a five-pointed star with a hammer and sickle (for products produced before 1994) or a female head in a kokoshnik with a profile turned to the right for products of a later release, as shown in the table below.
The presence of an identification mark indicates that it has been assayed and stamped by the Assay Supervision Inspectorate.
The shape of the mark depends on the type of noble alloy. Below are sketches of hallmarks:
- pos. 1 – for gold items (in the form of a kind of spatula);
- pos. 2 – for silver alloys (barrel-shaped);
- pos. 3 – for products made of platinum (in the form of a polyhedron);
- pos. 4 – for products made of palladium (truncated cone with a semicircular base).
The contents of the hallmark indicate the following information (for example, the image of the hallmark of a 750-karat gold product is used and is indicated in red letters):
- pos. A – code of the supervision inspection;
- pos. B – identification mark;
- pos. B – test sign.
What is a sample?
Fineness is the quantitative content of pure precious metal in a jewelry alloy. The sample indicator is the mass of the noble metal expressed in grams , which is contained in 1 kg of the alloy.
The sample is assigned to any jewelry alloy.
For jewelry and household products made of precious metals in the Russian Federation in accordance with the Decree of the Government of the Russian Federation of June 18, 1999. The following samples were accepted for No. 643:
- for gold – 375, 500, 585, 750, 958 and 999;
- for silver – 800, 830, 875, 925, 960 and 999;
- for platinum – 850, 900 and 950;
- for palladium – 500 and 850.
Assay marks on jewelry are usually placed in the following places :
- for rings, use the inner side of the rim;
- for pendants, use the outer side of the ear;
- lock details are used on earrings, brooches and chains.
Which metals are noble and their properties?
This group of metals received the name “noble” due to their special characteristics. Depending on the variety, their physical and chemical properties may manifest themselves to varying degrees, but they always remain unique.
Rhodium
Rhodium is a member of the platinum group. It is a light metal and has a pale blue color. It is distinguished by a high degree of hardness and, at the same time, fragility.
Valued for its high reflectivity and resistance to chemical attack. Rhodium can only be oxidized with hot sulfuric acid. The melting process begins when heated to almost 2000 °C.
Platinum
Because of its white luster, platinum, discovered in the mines of America, was originally called “silver.” Only in 1751 platinum received the status of a precious metal, and its value instantly surpassed the then known silver and gold. It has high ductility and can be easily forged (which is why it is loved by jewelers). At the same time, platinum is harder than gold, refractory, resistant to chemical influences, and not subject to oxidation.
Gold
Like platinum, it has good ductility and malleability, but has lower melting points. Reacts only with aqua regia, invulnerable to alkalis, salts and acids. In nature, specimens of pure gold with a pronounced yellow color and characteristic shine are rarely found. Most often, miners encounter green faded ore.
Osmium
The most refractory of the noble metals. The melting point reaches 2700 °C. In addition, osmium does not dissolve in acids. External characteristics are white and hard. Belongs to the group of heavy metals.
Iridium
Like osmium, it is a heavy metal. The most durable, dense, refractory and insoluble in acids, gray-white in color. The melting point is slightly lower than that of osmium and is 2454 °C.
Ruthenium
In terms of external characteristics, ruthenium can easily be confused with platinum. The noble metal resembles iridium in its melting point and has increased strength and density. It is interesting that only ruthenium and osmium form water-soluble cakes under the influence of alkali, oxidizing agent and high temperatures.
Palladium
Soft, malleable, white with a silver tint. When heated to 860°C, palladium forms oxides, but becomes pure again as the temperature rises further. The melting point is 1554 °C.
Silver
Among the noble metals, silver has the lowest density and a relatively low melting point - 960 ° C. It is best forged and serves as an excellent heat and electrical conductor. It practically does not react with acids, but darkens under the influence of hydrogen sulfide, which is part of the atmosphere.
List of semi-precious metals
In jewelry production and instrument making, metals are actively used that are not essentially precious, but have a certain value. They are conventionally called semi-precious. Among the most popular types are the following:
- titanium;
- tungsten;
- cupronickel
Their price fluctuates in the average price range and does not exceed $2 per gram.
Current state of primary production
Gold mining occupies a priority position in the mining industry of any country.
A distinctive feature of modern primary mining of precious metals is the depletion of almost all known deposits .
For gold mining, industrial processing of ore from a primary deposit is recognized as profitable when extracting gold in the range of 1-5 g per ton of rock, and for placer mines the content of 0.1-0.5 g of gold grains per ton of processed sand is already welcomed and is considered beneficial for development.
quite large undeveloped gold deposits in Russia .
Against the backdrop of problems associated with the practical implementation of technologies for primary mining and processing of millions of tons of rock for the sake of just a few kilograms of gold or platinum, methods for processing metal-containing secondary raw materials are gaining popularity.
Scrap of precious metals and waste from the production of products containing noble metals are hundreds and thousands of times higher than natural sources in terms of the content of noble components in them. You can read about the cost of scrap precious metals in this article.
In many industrialized countries, recycling technologies return precious metals in quantities of up to 40% of the total volume of gold and other precious metals produced.
Despite the obvious stagnation of primary gold production, the level of its production, like other precious metals, is growing year after year. This is due to an increase in the volume of secondary production of precious metals.
History of appearance
Noble metals are a valuable and non-renewable natural resource. None of the precious metals can be obtained experimentally, so for the scientific community the appearance of these elements on Earth still remains a mystery. At the moment, there are two working versions of their appearance:
- Space. Proponents of this hypothesis believe that we owe the appearance of precious metals in the earth's crust to meteorites that bombarded the Earth at the dawn of its formation. However, this version has a serious drawback - scientists have found that the average meteorite contains only about 0.005% of the total weight of noble metals, which in no way correlates with the volumes mined at different deposits.
- Tectonic. According to this assumption, all precious metals were born under the influence of high temperatures and in the presence of special conditions in the core of the planet, and then were thrown to the surface with lava flows. This version has more supporters than the space version, however, it cannot answer all the questions. In particular, it is not clear why these natural resources stopped forming again and entering the upper layers of the earth's crust along with volcanic lava emissions.
The question of the origin of noble metals and their alloys is one of the most important. If scientists are ever able to answer it, it will change the entire existing system of relationships in the world.
Sources of secondary raw materials
Sources of precious secondary raw materials can be:
- failed parts of industrial equipment, measuring equipment and control automation;
- waste from the production of radio electronics, television equipment, chemical industry, metallurgy, printing and other industrial sectors;
- jewelry production waste;
- scrap of precious metals.
Scrap of precious metals, considered as secondary raw materials, is represented by the following main groups of used products :
- For gold and silver scrap, the sources are used jewelry, thermocouples and temperature sensors, parts of household refrigerators, televisions, elements of office equipment (computers and printers), radio components, gadgets, telecommunications equipment, catalysts and catalytic grids, batteries, etc.
- For platinum group metal scrap, sources include jewelry made from various alloys of platinum and palladium, catalytic meshes and thermocouples, electrolysis equipment, laboratory glassware, crucibles and lids for them, other laboratory supplies, cases of glass melting equipment, dies, fragments of products coated with precious metals .
Rhodium
This element has a bluish tint. Like argentum, it is reflective, and rhodium is particularly hard and brittle. Otherwise, all its qualities coincide with the properties described above, characteristic of all noble metals. Rhodium is slightly denser than palladium - twelve point one and forty-four hundredths of a gram per cubic centimeter. As for the melting and hardening temperature, this element softens at a temperature of 1966 ° C (even more than platinum!), and takes a solid form at 6 Mohs. The physical and chemical properties of rhodium allow it to be used to coat silver and gold items.
Content of precious metals in secondary raw materials
By its chemical composition, secondary raw materials of precious metals are complex multicomponent metallurgical raw materials.
As a rule, in addition to precious metals, scrap and waste contain alloys of iron, aluminum and copper, glass, glass ceramics, fiberglass, and polymers.
The content of precious metals in various objects and devices can only be determined after they have been processed in a refining organization capable of purifying gold, platinum, palladium and other metals to the highest standard.
The status of refining organizations is assigned to enterprises that carry out industrial purification (French affinage - purification, purification) of impurities of precious metals and bringing their quality to the requirements of the relevant standards.
to assess the quantitative content of gold, silver or other noble element in the “secondary” material prepared for processing on the basis of reference data that is available for each product or group of identical parts, or by a hallmark stamp on a piece of scrap jewelry.
For example, for a transistor of the KT 203 B series, the reference book indicates that it contains 0.0076 g of gold as gold plating on the terminals.
We offer you several articles on the topic of the content of precious metals in various products and their refining:
- Content of precious metals in radio components.
- Gold content in radio components.
- Contents in cars.
- Precious metals in household and office equipment.
- Refining of radio components.
- Refining of various items.
- Gold refining.
Platinum
This metal is in second place in value, it has been known since ancient times (Ancient Greece, Egypt, South America), but it was introduced as a chemical element by the chemist Schaeffer in 1952. Platinum is a rare precious silver-white metal that occurs in nature exclusively as a natural alloy with other metals. The process of obtaining pure metal is extremely labor-intensive and expensive. Platinum has high chemical resistance, resistance to oxidation in air, and acid resistance - the metal is not affected by acids.
Positive aspects of recycling
The main positive aspects of using the so-called “secondary” precious metals are the following circumstances :
- Unlike natural mineral raw materials used for primary mining and extraction of precious metals, secondary raw materials, represented by scrap gold, silver or platinum group metals, are much richer in the content of precious metals.
- Due to the increasing growth in the consumption of computer equipment, radio and television equipment, various gadgets, office equipment and household electrical appliances, the number of those that fail or are rejected during production or warranty service, which inherently serve as an endless source of precious metals in the form of recyclable materials, is correspondingly increasing. At the same time, the painful issue of recycling monitors, televisions, computers and other products that are subject to disposal in specialized organizations is being resolved.
- Precious metals recycling technologies do not require large production areas and have fewer technological stages compared to primary mining.
The gold content in secondary raw materials, depending on the type of scrap, can range from 1% to 50% by weight; for silver the figure is somewhat more modest - from 0.1% to 15%. This means that 100-200 kg or more of the precious metal can be extracted from 1 ton of gold scrap.
Where are they used?
Immunity to external influences, aesthetics, plasticity, and other useful utilitarian characteristics have made “precious stones” a material for beautiful things, medicine, and high technology.
Beauty sphere
Gold, silver, platinum are traditional jewelry metals. Unlike platinum as the base material of jewelry, they are only plated with rhodium. The result of rhodium plating is that yellow gold looks more aristocratic white, and the strength threshold increases.
Gold jewelry
This is a decorative material (especially silver). Candelabra, boxes, powder compacts are pleasing to the eye and make life more enjoyable.
Silver ring with onyx
Industry
Industrialists have assessed the heat resistance and chemical inertness of materials:
- Gold, rhodium, silver – electroplating of circuit boards and contacts.
- This is the basis of solder, the filling of especially dense batteries, and the material of thermocouples.
- Ligature for common metals.
- Component of thermoelements, relay contacts, and other products.
- Platinoids are a mandatory ingredient in equipment for working in extreme conditions.
- In the chemical industry it is a material for laboratory containers, a catalyst.
- The military demands precious metals as a component of ammunition, lasers, and bomb casings.
Plastic surgery has developed the technology of sewing gold “rejuvenating” threads under the skin.
For scientists, gold is a material in the process of exploring the potential of radioactive materials.
Finance
The main area of application of precious metals is banking and financial:
- Gold, platinum, and palladium are classified as currency values: the central banks of many countries classify them as reserves along with currency and use them to pay each other.
- This is a tool for private investors. Especially in the form of bars and coins that are minted by the National Banks of many countries.
Silver coins
Precious metal coins are a proven way to store capital.
The world leader in the production of palladium is Russia, platinum is South Africa.